Online Database of Chemicals from Around the World
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical reagent >> Organic reagent >> Urea |
|---|---|
| Name | 4-Ethyl-3-thiosemicarbazide |
| Synonyms | 1-amino-3-ethylthiourea |
| Molecular Structure | 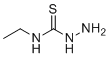 |
| Molecular Formula | C3H9N3S |
| Molecular Weight | 119.19 |
| CAS Registry Number | 13431-34-0 |
| EC Number | 236-553-1 |
| SMILES | CCNC(=S)NN |
| Density | 1.142±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 82-84 °C (Expl.) |
| Index of Refraction | 1.564, Calc.* |
| Boiling Point | 187.43±23.0 °C (760 mmHg), Calc.* |
| Flash Point | 67.159±22.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
4-Ethyl-3-thiosemicarbazide (C3H9N3S) is an organic compound belonging to the thiosemicarbazide family, characterized by a thioamide functional group and a hydrazine derivative structure. This chemical has found utility in a variety of industrial and biochemical applications due to its reactivity and ability to form derivatives with different chemical species. The discovery of 4-ethyl-3-thiosemicarbazide can be traced back to early 20th-century research on thiosemicarbazides, which were initially synthesized for their roles in organic synthesis and as intermediates for complex chemical transformations. The compound’s structure, featuring an ethyl substitution at the nitrogen atom, distinguishes it from other thiosemicarbazides, conferring unique reactivity and biological properties. One prominent application of 4-ethyl-3-thiosemicarbazide lies in the field of analytical chemistry. Due to its ability to form stable complexes with metal ions, it has been used as a chelating agent in qualitative and quantitative metal ion detection. Its reactivity with carbonyl groups also makes it a useful reagent in organic synthesis, facilitating the formation of heterocyclic compounds, such as thiazoles and triazoles. These heterocycles are essential precursors in the development of pharmaceuticals and agrochemicals. In medicinal chemistry, 4-ethyl-3-thiosemicarbazide has gained attention for its antimicrobial and antiproliferative properties. Studies have shown that derivatives of this compound exhibit inhibitory activity against bacterial and fungal pathogens. Additionally, some thiosemicarbazide derivatives demonstrate cytotoxic activity, making them candidates for further development as anticancer agents. This has spurred research into structure-activity relationships to optimize its pharmacological potential. The compound has also found applications in polymer chemistry. It serves as a cross-linking agent in polymer networks, contributing to the mechanical and thermal properties of materials. This application is particularly valuable in the development of coatings, adhesives, and other industrial materials where enhanced durability is required. Environmental applications of 4-ethyl-3-thiosemicarbazide include its use in wastewater treatment. The compound can act as a scavenger for heavy metals, facilitating their removal from industrial effluents. This utility aligns with growing demands for sustainable chemical practices and environmental conservation. Despite its versatile applications, the use of 4-ethyl-3-thiosemicarbazide must be handled with care due to its potential toxicity. Safe handling practices and proper disposal protocols are critical to minimizing health and environmental risks. Future research aims to explore safer derivatives and assess its broader applications, particularly in green chemistry and sustainable materials. The versatility of 4-ethyl-3-thiosemicarbazide underscores its importance in both industrial and scientific domains. Its role as a building block for more complex molecules and materials ensures its continued relevance, while ongoing research promises to unlock new potential applications. References 2010. 4-Ethyl-1-(4-methyl-benzylidene)thiosemicarbazide. Acta Crystallographica Section E, 66(6). DOI: 10.1107/s1600536810017988 2018. Biological aspects of Schiff base-metal complexes derived from benzaldehydes: an overview. Journal of the Iranian Chemical Society, 15(12). DOI: 10.1007/s13738-018-1411-2 2024. Optimisation of the corrosion inhibition performance of isatin 4-ethyl-3-thiosemicarbazone for mild steel in sulfuric acid medium using response surface methodology. Chemical Papers, 78(11). DOI: 10.1007/s11696-024-03603-2 |
| Market Analysis Reports |