Online Database of Chemicals from Around the World
| Tyger Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.tygersci.com | |||
 | +1 (609) 434-0144 | |||
 | +1 (609) 434-0143 | |||
 | sales@tygersci.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2008 | ||||
| TH Chemica Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.thchemica.com | |||
 | +86 (573) 8283-5115 +86 13998887555 | |||
 | +86 (573) 8396-5652 | |||
 | thchemica@126.com info@thchemica.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shijiazhuang C-element Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.c-elementbiotech.com | |||
 | +86 18033710157 | |||
 | Lisa@C-elementBiotech.com | |||
 | QQ Chat | |||
 | WeChat: 18033710157 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Reddy Chemtech Inc. | USA | |||
|---|---|---|---|---|
 | www.reddychemtech.com | |||
 | +1 (404) 483-6270 | |||
 | +1 (770) 420-0884 | |||
 | sales@reddychemtech.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Aminopyridine |
|---|---|
| Name | 2-Aminonicotinamide |
| Synonyms | 2-Amino-3-pyridinecarboxamide |
| Molecular Structure | 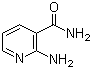 |
| Molecular Formula | C6H7N3O |
| Molecular Weight | 137.14 |
| CAS Registry Number | 13438-65-8 |
| EC Number | 623-981-2 |
| SMILES | C1=CC(=C(N=C1)N)C(=O)N |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 194-200 °C (Expl.) |
| Index of Refraction | 1.644, Calc.* |
| Boiling Point | 328.2±22.0 °C (760 mmHg), Calc.* |
| Flash Point | 152.3±22.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H317-H319 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P272-P280-P301+P316-P302+P352-P305+P351+P338-P321-P330-P333+P317-P337+P317-P362+P364-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2-Aminonicotinamide, a derivative of nicotinamide, is an organic compound with the molecular formula C6H7N3O. It consists of a pyridine ring substituted at the 2-position with an amino group and at the 3-position with a carboxamide group. The compound is structurally related to nicotinic acid and its amide form, nicotinamide, both of which are known for their biological relevance, especially as components of the vitamin B3 complex. The synthesis of 2-aminonicotinamide has been reported in multiple studies through well-established procedures. A common synthetic approach involves the transformation of 2-aminonicotinic acid or its ester into the corresponding amide via amidation reactions. The carboxylic acid group can be activated using reagents such as thionyl chloride or phosphorus pentachloride, followed by reaction with ammonia to yield the target compound. Alternatively, the ester form of 2-aminonicotinic acid can be directly aminolyzed with aqueous or gaseous ammonia to afford 2-aminonicotinamide in high yield. In chemical and pharmaceutical research, 2-aminonicotinamide is recognized for its role as an intermediate in the synthesis of heterocyclic compounds. Its functional groups enable participation in a variety of reactions, including condensation, acylation, and cyclization. The amino and amide groups are nucleophilic and form stable linkages with electrophilic reagents, making the compound suitable for constructing larger molecular frameworks. These properties have made it a valuable substrate for the development of pyridine-based compounds with potential pharmacological activity. 2-Aminonicotinamide has been studied in the context of biological applications, particularly as a scaffold for designing inhibitors of specific enzymes. Derivatives of this compound have been evaluated for activity against enzymes involved in cell proliferation, inflammation, and microbial metabolism. Its pyridine core and functional substituents are considered favorable for binding interactions in biological systems. Several analogs have demonstrated antibacterial, antifungal, and anticancer properties in in vitro screening programs, although 2-aminonicotinamide itself is primarily used as a synthetic precursor rather than a direct therapeutic agent. In addition to pharmaceutical applications, 2-aminonicotinamide serves as a useful ligand in coordination chemistry. The nitrogen atoms in the amino and pyridine moieties can chelate with metal ions, allowing the formation of coordination complexes. Such complexes have been investigated for their structural and catalytic properties. Metal complexes of 2-aminonicotinamide with transition metals like copper(II), zinc(II), and nickel(II) have been prepared and characterized using techniques such as infrared spectroscopy, UV-visible spectroscopy, and single-crystal X-ray diffraction. Analytical characterization of 2-aminonicotinamide includes standard spectroscopic and chromatographic methods. In infrared spectroscopy, it exhibits bands corresponding to N–H stretching from both the amino and amide groups, as well as a strong C=O stretching band typical of the amide functionality. Proton and carbon-13 nuclear magnetic resonance spectroscopy confirm the substitution pattern on the pyridine ring and verify the integrity of the functional groups. Mass spectrometry data provide molecular weight confirmation and fragmentation patterns consistent with the expected structure. The compound is typically handled as a solid and is stable under normal laboratory storage conditions. It is soluble in polar solvents such as water, ethanol, and dimethyl sulfoxide, which facilitates its use in synthetic transformations and analytical procedures. Safety data indicate that it should be handled with standard laboratory precautions, including protective clothing and eye protection. Overall, 2-aminonicotinamide remains an important compound in organic synthesis and medicinal chemistry, where its reactivity and structural properties continue to support the development of new molecular entities for research and therapeutic exploration. References 2020. Efficient Synthesis of Pyrido[2,3-d]pyrimidines by Recyclization of N-Arylitaconimides with Aminopyrimidinones. Russian Journal of Organic Chemistry, 56(9). DOI: 10.1134/s107042802009002x 2016. In-Water Synthesis of Quinazolinones from 1,1-Dichloro-2-nitroethene and Anthranilamides. Synlett, 27(12). DOI: 10.1055/s-0035-1561640 2011. Substituted 2-aminonicotinamides in the synthesis of pyrido[2,3-d]pyrimidin-4(1H)-ones, 2,3-dihydropyrido- [2,3-d]pyrimidin-4(1H)-ones, and 11b,12-dihydropyrido- [2',3':4,5]pyrimido[2,1-a]isoindole-5,7-diones. Chemistry of Heterocyclic Compounds, 47(3). DOI: 10.1007/s10593-011-0756-6 |
| Market Analysis Reports |