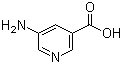
5-Aminonicotinic acid is an aminopyridinecarboxylic acid that belongs to the class of substituted pyridines, characterized by a pyridine ring bearing both an amino group and a carboxylic acid moiety. In the specific case of 5-aminonicotinic acid, the amino group is located at the 5-position relative to the carboxylic acid at the 3-position of the pyridine ring. The molecular formula of the compound is C6H6N2O2 and its structure contributes to its chemical reactivity and role in synthetic chemistry.
The synthesis of 5-aminonicotinic acid has been described in the literature since the mid-20th century. Classical synthetic routes involve nitration of nicotinic acid derivatives followed by reduction of the resulting nitro compounds to yield the corresponding amino acid. Specific synthetic approaches include the nitration of 3-cyanopyridine or 3-carboxypyridine under controlled conditions to introduce a nitro group at the 5-position, which is subsequently reduced to the amino group using hydrogenation or metal-based reducing agents. These methods are widely reported and have been optimized for yield and selectivity.
5-Aminonicotinic acid has attracted attention as a versatile intermediate in the synthesis of pharmaceutical and agrochemical compounds. The presence of both the amino and carboxyl functional groups enables it to participate in a variety of chemical reactions, including amidation, esterification, and diazotization. As a building block, it has been used in the synthesis of heterocyclic compounds, which are important in the development of biologically active molecules.
One of the notable applications of 5-aminonicotinic acid is its use in the preparation of compounds with potential pharmacological properties. It has been employed in the synthesis of analogs of nicotinamide and other pyridine-containing scaffolds, which are common motifs in many therapeutic agents. The compound has also been used as a precursor in the development of ligands for coordination chemistry and metal complex formation. In particular, its ability to coordinate with metal centers through the nitrogen atoms of the pyridine ring and amino group has been studied in the context of complex formation with transition metals.
In analytical chemistry, 5-aminonicotinic acid and its derivatives have been utilized in methods involving derivatization reactions to enhance detection sensitivity. The amino group can be modified with various reagents to introduce chromophores or fluorophores, allowing for application in spectrophotometric or chromatographic analysis.
The compound has also been investigated as a potential corrosion inhibitor. Aminopyridine derivatives, including 5-aminonicotinic acid, have shown the ability to adsorb onto metal surfaces and form protective films that reduce corrosion rates in acidic media. These studies have focused on the electrochemical behavior of the compound and its interaction with metal substrates such as steel.
From a physicochemical standpoint, 5-aminonicotinic acid is a crystalline solid, soluble in water and polar solvents due to the hydrophilic nature of its functional groups. Its acid-base properties, such as pKa values, have been studied in aqueous solution and are important for understanding its behavior in different pH environments, which is relevant for both synthetic applications and biological compatibility.
The compound is commercially available and has been cataloged in chemical databases for research and industrial use. Its known stability, reactivity, and synthetic accessibility make it a useful compound in the toolkit of synthetic and medicinal chemists. Its applications, as documented in the literature, are well-characterized and reflect its utility in multiple fields of chemical science.
References
2021. Preparation and Evaluation of Colon-Targeted Prodrugs of the Microbial Metabolite 3-Indolepropionic Acid as an Anticolitic Agent. Molecular Pharmaceutics, 18(3).
DOI: 10.1021/acs.molpharmaceut.0c01228
2021. 5-[(3-Carboxy-4-hydroxyphenyl)diazenyl] nicotinic acid, an azo-linked mesalazine-nicotinic acid conjugate, is a colon-targeted mutual prodrug against dextran sulfate sodium-induced colitis in mice. Journal of Pharmaceutical Investigation, 51(3).
DOI: 10.1007/s40005-021-00517-z
2020. Structural elucidation, molecular docking, α-amylase and α-glucosidase inhibition studies of 5-amino-nicotinic acid derivatives. BMC Chemistry, 14(1).
DOI: 10.1186/s13065-020-00695-1
|















































































 GHS07 Warning Details
GHS07 Warning Details