Online Database of Chemicals from Around the World
| Xi'an Tuochao Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xatuochao.cn | |||
 | +86 18092957403 | |||
 | chen.zeshan@xatuochao.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: +8618092957403 | |||
 | WhatsApp:+8618092957403 | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Methyl 3-bromo-6-chloropyrazine-2-carboxylate |
| Molecular Structure | 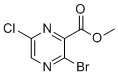 |
| Molecular Formula | C6H4BrClN2O2 |
| Molecular Weight | 251.47 |
| CAS Registry Number | 13457-28-8 |
| SMILES | COC(=O)C1=NC(=CN=C1Br)Cl |
| Density | 1.8±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 35-36 °C |
| Index of Refraction | 1.574, Calc.* |
| Boiling Point | 292.4±35.0 °C (760 mmHg), Calc.* |
| Flash Point | 130.6±25.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
Methyl 3-bromo-6-chloropyrazine-2-carboxylate is an important chemical compound within the field of medicinal chemistry and agrochemicals, known for its diverse applications in synthesis and research. Its molecular formula is C8H6BrClN2O2, and it features both halogen atoms and a carboxylate group, which contributes to its reactivity and utility in various chemical transformations. The discovery of methyl 3-bromo-6-chloropyrazine-2-carboxylate can be traced back to research focused on the synthesis of pyrazine derivatives, which are recognized for their biological activity and structural complexity. Pyrazine and its derivatives have been investigated for their potential as pharmaceutical agents, fungicides, and herbicides. The introduction of halogen substituents, such as bromine and chlorine, in the pyrazine ring enhances its reactivity and allows for further functionalization, making it a valuable building block in organic synthesis. One of the notable applications of methyl 3-bromo-6-chloropyrazine-2-carboxylate lies in the synthesis of biologically active compounds. The presence of the bromine and chlorine atoms facilitates cross-coupling reactions, such as Suzuki and Sonogashira reactions, which are widely used to construct complex organic molecules. Additionally, the compound has been explored for its potential use in the development of new agrochemicals, particularly in designing novel herbicides and fungicides that can target specific plant pathogens. Research has demonstrated that modifications of the pyrazine ring can lead to compounds with significant pharmacological properties. For instance, derivatives of methyl 3-bromo-6-chloropyrazine-2-carboxylate have been synthesized and evaluated for their activity against various cancer cell lines, showcasing the potential of such compounds in medicinal chemistry. Furthermore, the compound serves as an intermediate in the synthesis of more complex molecules, allowing researchers to explore structure-activity relationships and optimize compounds for specific biological activities. In summary, methyl 3-bromo-6-chloropyrazine-2-carboxylate is a significant compound in organic synthesis and drug development, with applications in both medicinal chemistry and agrochemicals. Its unique structure, featuring halogen substituents, provides a versatile platform for further chemical modifications and research into novel therapeutic agents. References none |
| Market Analysis Reports |