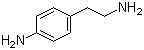
2-(4-Aminophenyl)ethylamine, also known as 4-Amino-β-phenethylamine, is a chemical compound with the molecular formula C8H12N2 and a structural formula that features an ethylamine moiety attached to a para-aminophenyl group. This compound has garnered interest in both medicinal chemistry and organic synthesis due to its unique structural properties and potential biological activities.
The discovery of 2-(4-aminophenyl)ethylamine can be traced back to early research on phenethylamines, which are known to possess various pharmacological effects. The para-amino substitution on the phenyl ring plays a significant role in the compound's biological activity, as it can influence receptor interactions and modulate various physiological responses. The compound is considered a derivative of phenethylamine, a well-known scaffold in medicinal chemistry, with a range of biological activities, including stimulant effects and potential therapeutic applications.
One of the primary applications of 2-(4-aminophenyl)ethylamine lies in its use as a precursor in the synthesis of various pharmaceutical agents. It can serve as an intermediate for producing compounds with analgesic, anti-inflammatory, and stimulant properties. For instance, derivatives of 2-(4-aminophenyl)ethylamine have been investigated for their potential in treating conditions such as attention deficit hyperactivity disorder (ADHD) and depression, owing to their ability to interact with neurotransmitter systems in the brain.
Furthermore, the compound has been utilized in the development of novel ligands for various receptors, including adrenergic and dopaminergic receptors. This research has highlighted the potential of 2-(4-aminophenyl)ethylamine derivatives in drug discovery programs aimed at creating new therapeutics for neurological and psychiatric disorders. The ability to modify the amine and aromatic groups allows for the exploration of structure-activity relationships, leading to the identification of compounds with improved efficacy and selectivity.
In addition to its pharmaceutical applications, 2-(4-aminophenyl)ethylamine has been employed in organic synthesis as a building block for more complex molecules. Its versatility in reactions, such as alkylation and acylation, enables chemists to create diverse chemical entities, further expanding its utility in research and development.
In conclusion, 2-(4-aminophenyl)ethylamine is a significant compound in the fields of medicinal chemistry and organic synthesis, with applications ranging from drug development to organic reactions. Its structural features and biological potential make it a valuable subject of study in the quest for novel therapeutic agents.
References
2011. Rational and Predictable Chemoselective Synthesis of Oligoamines via Buchwald-Hartwig Amination of (Hetero)Aryl Chlorides Employing Mor-DalPhos. The Journal of Organic Chemistry, 77(1).
DOI: 10.1021/jo202358p
2016. Bacterial fermentation platform for producing artificial aromatic amines. Scientific Reports, 6.
DOI: 10.1038/srep25764
2022. Fermentative production of 2-(4-aminophenyl)ethylamine to synthesize a novel heat-resistant biopolyurea. Bioscience, Biotechnology, and Biochemistry, 86(7).
DOI: 10.1093/bbb/zbac077
|




























































 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details