Online Database of Chemicals from Around the World
| Neijiang Noble Materil Technology. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njsnoble.com | |||
 | +86 (832) 220-6225 | |||
 | +86 (832) 220-6225 | |||
 | 3080634228@qq.com | |||
| Chemical distributor since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Kangchi Chemical Industry (shenzhen) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.konchie.com | |||
 | +86 (755) 8257-4253 | |||
 | +86 (755) 3315-1032 | |||
 | kangchi@konchie.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| W.C.Heraeus GmbH | Germany | |||
|---|---|---|---|---|
 | www.heraeus-chemicalproducts.com | |||
 | +49 (6181) 355-707 | |||
 | +49 (6181) 355-914 | |||
 | info@heraeus.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal sulfides and sulfates |
|---|---|
| Name | Tetraamminepalladium sulfate |
| Synonyms | Azane palladium(2+) sulfate |
| Molecular Structure | 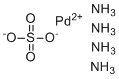 |
| Molecular Formula | Pd.(NH3)4.SO4 |
| Molecular Weight | 270.60 |
| CAS Registry Number | 13601-06-4 |
| EC Number | 426-980-3 |
| SMILES | N.N.N.N.[O-]S(=O)(=O)[O-].[Pd+2] |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H319 Details | ||||||||||||||||||||||||||||
| Safety Statements | P305+P351+P338 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Tetraamminepalladium sulfate is a coordination compound with the formula [Pd(NH3)4]SO4. It consists of a palladium(II) center coordinated by four ammonia (NH3) ligands, forming a cationic complex [Pd(NH3)4]2+, paired with the sulfate anion SO42– to balance charge. This compound typically forms crystalline solids that are soluble in water, reflecting the ionic nature of the sulfate salt and the polar character of the complex. The synthesis of tetraamminepalladium sulfate involves the reaction of palladium(II) salts, such as palladium chloride or palladium sulfate, with an excess of ammonia in aqueous solution. The ammonia molecules coordinate to the palladium ion via nitrogen lone pairs, replacing labile ligands such as chloride or water. This process yields a stable, square planar complex characteristic of d8 palladium(II) ions, where the palladium center is surrounded by four ammonia ligands arranged symmetrically. Structurally, the palladium center exhibits a square planar geometry with Pd–N bond lengths typical for palladium ammine complexes. The sulfate anions remain outside the coordination sphere, balancing the complex's positive charge. The compound crystallizes in various hydrated or anhydrous forms depending on conditions and drying methods. Tetraamminepalladium sulfate is mainly used as an intermediate and precursor in coordination chemistry and homogeneous catalysis. It serves as a versatile starting material for preparing a wide range of palladium complexes through ligand substitution reactions, allowing for the introduction of phosphines, nitrogen donors, or other ligands tailored to specific catalytic or synthetic needs. In catalysis, palladium complexes derived from tetraamminepalladium sulfate are employed in various organic transformations, including carbon–carbon coupling reactions (e.g., Suzuki, Heck reactions) that are fundamental in synthetic organic chemistry for constructing complex molecules. The tetraammine ligand framework provides stability while permitting ligand exchange, thus enabling the generation of catalytically active species under mild conditions. Beyond catalysis, tetraamminepalladium sulfate is also used in materials science for the preparation of palladium-containing films and nanoparticles. Controlled decomposition or reduction of the complex allows for the formation of palladium metal deposits, which are valuable in electronics, sensors, and hydrogen storage technologies. The compound is typically handled in aqueous solutions under standard laboratory safety practices. Like other palladium complexes, it should be managed with care to avoid exposure due to the potential toxicity and environmental impact of palladium compounds. In summary, tetraamminepalladium sulfate is a well-defined palladium(II) ammine complex used as a precursor in coordination chemistry and catalysis. It offers a stable yet reactive platform for generating diverse palladium catalysts and materials. Its preparation from palladium salts and ammonia is straightforward, and its structural features enable applications across synthetic chemistry and materials science. References none |
| Market Analysis Reports |