Online Database of Chemicals from Around the World
| Baoji Rock Pharmachem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bjrock.com | |||
 | +86 (10) 8487-1070 8487-1071 8487-1072 | |||
 | +86 (10) 8487-1078 | |||
 | bjrock@163.com | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shaanxi Kaida Chemcial Engineering Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kd-chem.com | |||
 | +86 (917) 331-7143 332-2918 | |||
 | +86 (917) 331-0616 | |||
 | kaida@kd-chem.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Kangchi Chemical Industry (shenzhen) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.konchie.com | |||
 | +86 (755) 8257-4253 | |||
 | +86 (755) 3315-1032 | |||
 | kangchi@konchie.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Heraeus Ltd. | Hong Kong | |||
|---|---|---|---|---|
 | www.heraeus.hk | |||
 | +852 2675-1200 2773-1733 | |||
 | +852 2773-1090 / 2682-3220 | |||
 | cpd@heraeus.com.hk | |||
| Chemical manufacturer | ||||
| W.C.Heraeus GmbH | Germany | |||
|---|---|---|---|---|
 | www.heraeus-chemicalproducts.com | |||
 | +49 (6181) 355-707 | |||
 | +49 (6181) 355-914 | |||
 | info@heraeus.com | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Colonial Metals, Inc. | USA | |||
|---|---|---|---|---|
 | www.colonialmetals.com | |||
 | +1 (410) 398-7200 (800) 962-1537 | |||
 | +1 (410) 398-2918 | |||
 | sales@colonialmetals.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Inorganic ammonium salt |
|---|---|
| Name | Tetraammineplatinum(II) chloride |
| Synonyms | Tetraammineplatinum dichloride |
| Molecular Structure | 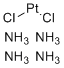 |
| Molecular Formula | PtCl2.(NH3)4 |
| Molecular Weight | 334.11 |
| CAS Registry Number | 13933-32-9 |
| EC Number | 237-706-5 |
| SMILES | Cl[Pt]Cl.N.N.N.N |
| Hazard Symbols |     GHS05;GHS07;GHS08;GHS09 Danger Details GHS05;GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H290-H302-H315-H317-H318-H335-H410 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P280-P305+P351+P338-P310 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 3077 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Tetraammineplatinum(II) chloride is a coordination compound with the chemical formula [Pt(NH3)4]Cl2. It consists of a platinum(II) ion coordinated by four ammonia (NH3) ligands forming the cationic complex [Pt(NH3)4]2+, paired with two chloride anions to balance the charge. This compound is a well-characterized example of a platinum(II) ammine complex and has been studied extensively for its chemical properties and applications. The synthesis of tetraammineplatinum(II) chloride typically involves the reaction of platinum(II) chloride or other platinum(II) precursors with an excess of aqueous ammonia. Under controlled conditions, ammonia molecules coordinate to the platinum center by donating lone pairs of electrons on nitrogen, displacing other ligands if present. The resulting complex adopts a square planar geometry characteristic of d8 platinum(II) ions, with the four ammonia ligands arranged symmetrically around the platinum atom. Structurally, the platinum center is coordinated by four ammonia ligands in a square planar configuration. The platinum–nitrogen bond lengths and ligand arrangement conform to well-established principles of coordination chemistry. The chloride ions remain outside the coordination sphere as counterions to maintain overall electrical neutrality. The compound typically forms crystalline solids that are soluble in water due to its ionic nature. Tetraammineplatinum(II) chloride has been used as a precursor in coordination chemistry for the preparation of various platinum complexes. Its ammonia ligands can be substituted partially or fully by other ligands such as phosphines, nitrogen heterocycles, or sulfur donors, enabling the synthesis of complexes with tailored properties for catalysis, medicinal chemistry, and materials science. In catalysis, platinum complexes derived from tetraammineplatinum(II) chloride can serve as homogeneous catalysts in hydrogenation, hydrosilylation, and other organometallic transformations. The relatively stable ammine coordination environment facilitates ligand exchange under controlled conditions, allowing modulation of catalytic activity and selectivity. While the related platinum(II) complex cisplatin is renowned for its anticancer properties, tetraammineplatinum(II) chloride itself is not typically used as a drug but rather as an intermediate in synthetic chemistry. It provides a convenient and well-defined platform for preparing other biologically active platinum complexes. Handling of tetraammineplatinum(II) chloride requires standard laboratory precautions. It is generally stable under normal conditions but should be stored away from strong acids and reducing agents to prevent decomposition. As with other platinum compounds, care should be taken to avoid inhalation or ingestion due to potential toxicity. In summary, tetraammineplatinum(II) chloride is a square planar platinum(II) complex with four coordinated ammonia ligands and two chloride counterions. It is synthesized by coordination of ammonia to platinum precursors and is widely used as a precursor in platinum coordination chemistry and catalysis. Its stability, well-defined structure, and ligand substitution capability make it a valuable compound in chemical research and development. References none |
| Market Analysis Reports |