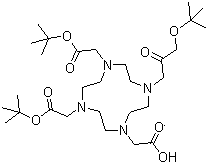
Tri-tert-butyl 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetate is a highly substituted macrocyclic ligand featuring a tetraazacyclododecane core structure with four acetato groups at the 1, 4, 7, and 10 positions of the ring. This compound, which belongs to a class of chelating ligands known as cyclen derivatives, is of significant interest in the fields of coordination chemistry, materials science, and medicinal chemistry.
The central macrocyclic ring in tri-tert-butyl 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetate consists of four nitrogen atoms and a 12-membered carbon ring. This structure allows the molecule to coordinate with metal ions, forming stable metal-ligand complexes. The acetato groups at the 1, 4, 7, and 10 positions serve as donor sites for metal coordination, providing flexibility and increasing the stability of the complex formed with the metal ions. The tert-butyl groups attached to the nitrogen atoms further enhance the steric bulk of the ligand, which can influence the reactivity and selectivity of metal ion coordination.
Synthesis of this ligand typically involves the reaction of 1,4,7,10-tetraazacyclododecane (often referred to as cyclen) with acetic anhydride or acetyl chloride to introduce the acetato groups at the desired positions. The tert-butyl groups are incorporated into the molecule by reacting the cyclen derivative with tert-butyl iodide or a similar reagent, allowing for the selective introduction of bulky substituents that contribute to the steric and electronic properties of the final compound.
Tri-tert-butyl 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetate has broad applications in coordination chemistry and materials science. The ligand’s ability to form stable complexes with metal ions makes it valuable in the design of metal-based catalysts for a range of chemical reactions, including oxidation, reduction, and polymerization processes. The steric protection offered by the tert-butyl groups helps to ensure selective binding and prevents the formation of undesired coordination complexes with smaller metal ions.
In addition to its role in catalysis, this ligand has potential applications in the field of medicinal chemistry. Metal complexes of cyclen-based ligands are often studied for their ability to act as imaging agents or therapeutic agents. The stability and solubility of the complexes, which can be tailored by varying the nature of the ligand, make them promising candidates for use in medical imaging, particularly in magnetic resonance imaging (MRI) and positron emission tomography (PET). Metal-ligand complexes can also be used in the delivery of drugs to targeted tissues, taking advantage of the coordination properties of the ligand to transport therapeutic agents to specific sites in the body.
The coordination properties of tri-tert-butyl 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetate also make it a valuable intermediate in the synthesis of new materials with specific electronic or optical properties. For example, complexes of this ligand with transition metals may exhibit interesting magnetic or photonic properties that can be utilized in the development of novel materials for sensors, electronics, or light-emitting devices.
In conclusion, tri-tert-butyl 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetate is a versatile ligand with a wide range of potential applications in coordination chemistry, catalysis, and medicinal chemistry. Its ability to form stable complexes with metal ions, coupled with the steric protection provided by the tert-butyl groups, makes it a useful tool in the design of metal-based catalysts, imaging agents, and drug delivery systems. Further research into its properties and applications could lead to the development of new technologies in a variety of scientific fields.
|





































 GHS07 Warning Details
GHS07 Warning Details