Online Database of Chemicals from Around the World
| Zouping Mingxing Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zoutong.com.cn | |||
 | +86 13605431940 | |||
 | +86 (543) 224-0079 | |||
 | sdzpmxchem@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Survival Technologies Pvt Ltd | India | |||
|---|---|---|---|---|
 | www.survivaltechnologies.in | |||
 | +91 (22) 4212-8221 | |||
 | +91 (22) 4212-8226 | |||
 | info@survivaltechnologies.in | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shijiazhuang Lida Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lid-ff.cn | |||
 | +86 (311) 8573-7996 | |||
 | +86 (311) 8573-9175 | |||
 | service@lid-ff.cn | |||
| Chemical manufacturer since 1993 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Henan Tianfu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tianfuchem.com | |||
 | +86 (371) 5517-0693 | |||
 | +86 (371) 5517-0975 | |||
 | leon@tianfuchem.com | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Beijing Guofang Dingsheng Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.gfdschem.com | |||
 | +86 (10) 8904-9254 5909-2859 +86 18610472091 +86 18031679351 | |||
 | +86 (10) 5909-2859 | |||
 | gfdskj@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Gelest, Inc. | USA | |||
|---|---|---|---|---|
 | www.gelest.com | |||
 | +1 (215) 547-1015 | |||
 | +1 (215) 547-2484 | |||
 | info@gelest.com | |||
| Chemical manufacturer | ||||
| MCAT GmbH | Germany | |||
|---|---|---|---|---|
 | www.mcat.de | |||
 | +49 (7531) 939-097 | |||
 | +49 (7531) 939-098 | |||
 | info@mcat.de | |||
| Chemical manufacturer | ||||
| DeLong Chemicals America | USA | |||
|---|---|---|---|---|
 | www.delongchemicals.com | |||
 | +1 (203) 271-9017 | |||
 | sales@delongchemicals.com | |||
| Chemical manufacturer | ||||
| Dalchem | Russia | |||
|---|---|---|---|---|
 | www.dalchem.com | |||
 | +7 (8312) 753-772 | |||
 | +7 (8312) 750-799 | |||
 | dregichy@dalchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Amoy Cpharma Company Limited | China | |||
|---|---|---|---|---|
 | www.amoy-cpharma.com | |||
 | +86 (592) 228-3485 | |||
 | +86 (592) 228-3495 | |||
 | cpharma@public.xm.fj.cn | |||
| Chemical distributor | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | Tri-tert-butylphosphine |
| Synonyms | Tris(1,1-dimethylethyl)phosphine |
| Molecular Structure | 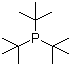 |
| Molecular Formula | C12H27P |
| Molecular Weight | 202.32 |
| CAS Registry Number | 13716-12-6 |
| EC Number | 237-266-4 |
| SMILES | CC(C)(C)P(C(C)(C)C)C(C)(C)C |
| Density | 0.82 |
|---|---|
| Melting point | 30 °C |
| Boiling point | 102-103 °C (13 mmHg) |
| Flash point | -17 °C |
| Water solubility | insoluble |
| Hazard Symbols |   GHS02;GHS05 Danger Details GHS02;GHS05 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H250-H314-H318 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P222-P231-P233-P260-P264-P264+P265-P280-P301+P330+P331-P302+P335+P334-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P363-P370+P378-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2846 | ||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Tri-tert-butylphosphine (TTBP) is an important organophosphorus compound that was first synthesized and characterized in the mid-20th century when chemists were exploring new phosphorus-based ligands and reagents. The synthesis of this compound involves the reaction of tert-butylmagnesium chloride with phosphorus trichloride, followed by purification to obtain pure TTBP. Its discovery marked a major advance in phosphine chemistry, providing enhanced steric protection and reactivity control in various chemical transformations. The molecular formula of tri-tert-butylphosphine is C₉H₁₈P, which consists of a phosphorus atom bonded to three tert-butyl groups (-C(CH₃)₃). This structure creates significant steric hindrance around the phosphorus center, affecting its reactivity and coordination behavior. TTBP is a colorless liquid at room temperature with a characteristic odor. It is soluble in nonpolar solvents such as benzene, toluene, and dichloromethane, facilitating organic reactions and solution phase chemistry. TTBP exhibits nucleophilic and reducing properties due to the electron-donating nature of the tert-butyl groups. It readily coordinates with transition metals and metalloids to form stable complexes for use in catalytic processes and semiconductor applications. TTBP complexes with transition metals such as palladium (Pd) or platinum (Pt) are effective catalysts for hydrogenation reactions. These catalysts promote the selective reduction of alkenes, alkynes, and carbonyl compounds under mild conditions and have advantages in synthetic organic chemistry. TTBP-based palladium catalysts are used in cross-coupling reactions to promote the formation of carbon-carbon bonds (e.g., Suzuki-Miyaura coupling). These reactions are essential in drug synthesis and materials science for building complex organic molecules and functional materials. TTBP is used as a precursor for the production of doped semiconductors. It helps in the incorporation of phosphorus atoms into silicon (Si) or germanium (Ge) matrices, thereby modifying their electronic properties for use in semiconductor devices. Doped semiconductors have enhanced conductivity and tailored electrical properties, which are essential for integrated circuits and electronic devices. TTBP-based compounds are used for surface passivation of semiconductor materials, mitigating surface defects and improving device performance. Passivation layers improve the stability and reliability of semiconductor devices, especially in light-emitting diodes (LEDs), solar cells, and microelectronic components. TTBP acts as a mild reducing agent in organic synthesis, facilitating the conversion of functional groups such as carbonyls (e.g., ketones, aldehydes) into the corresponding alcohols. The controlled reactivity of TTBP ensures high yields and selectivity in these transformations, supporting the production of pharmaceutical intermediates and fine chemicals. References 2023. Synthesis, crystal structure, and biological activity of menthol-based chiral quaternary phosphonium salts (CQPSs). Structural Chemistry. DOI: 10.1007/s11224-023-02259-0 2023. Miniaturization of popular reactions from the medicinal chemists’ toolbox for ultrahigh-throughput experimentation. Nature Synthesis. DOI: 10.1038/s44160-023-00351-1 2023. Design and Application of Novel Sterically Hindered Phosphonium Salts in the Development of Functional Materials. Russian Journal of General Chemistry, 93(17). DOI: 10.1134/s1070363223170036 2024. Thermal Decomposition of the Complex [tBu3PH][HB(C6F5)3]. Russian Journal of General Chemistry, 94(14). DOI: 10.1134/s1070363224140056 2024. Study of photophysical properties in bronsted acids for nitrogen atoms with different hybrid (sp, sp2, sp3) orbitals. Journal of the Iranian Chemical Society. DOI: 10.1007/s13738-024-03048-0 |
| Market Analysis Reports |