Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Alfachem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.alfachemch.com | |||
 | +86 (0371) 5505-2911 | |||
 | alfa5@alfachem.cn | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Combi-Blocks, Inc. | USA | |||
|---|---|---|---|---|
 | www.combi-blocks.com | |||
 | +1 (858) 635-8950 | |||
 | +1 (858) 635-8991 | |||
 | sales@combi-blocks.com | |||
| Chemical manufacturer | ||||
| Maybridge Chemical Co., Ltd. | Belgium | |||
|---|---|---|---|---|
 | www.maybridge.com | |||
 | +32 (14) 593-434 | |||
 | maybridge.sales@thermofisher.com | |||
| Chemical manufacturer | ||||
| Frinton Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.frinton.com | |||
 | +1 (856) 722-7037 | |||
 | +1 (856) 439-1977 | |||
 | sales@frinton.com frinton@frinton.com | |||
| Chemical manufacturer since 1963 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | 2,5-Dibromoterephthalic acid |
| Molecular Structure | 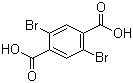 |
| Molecular Formula | C8H4Br2O4 |
| Molecular Weight | 323.92 |
| CAS Registry Number | 13731-82-3 |
| EC Number | 237-300-8 |
| SMILES | C1=C(C(=CC(=C1Br)C(=O)O)Br)C(=O)O |
| Density | 2.2±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 315-318 °C (Expl.) |
| Index of Refraction | 1.680, Calc.* |
| Boiling Point | 445.0±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 222.9±28.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
2,5-Dibromoterephthalic acid is an aromatic compound derived from terephthalic acid, characterized by bromine atoms substituted at the 2 and 5 positions of the benzene ring. This halogenated derivative plays a significant role in material science and organic synthesis, serving as a versatile intermediate for various applications. Its discovery emerged during research on functionalized terephthalic acids for advanced polymer and material development. The synthesis of 2,5-dibromoterephthalic acid typically involves bromination reactions of terephthalic acid under controlled conditions to achieve selective substitution. This compound's structure, combining bromine atoms and carboxylic acid groups, offers unique reactivity, making it a valuable precursor in diverse chemical transformations. 2,5-Dibromoterephthalic acid finds extensive use in the production of specialty polymers and advanced materials. It is utilized as a monomer in the synthesis of halogenated polyesters and polyamides, which exhibit enhanced thermal stability, flame retardance, and mechanical properties. These materials are critical in applications such as aerospace, electronics, and automotive industries. In addition to its role in polymer science, 2,5-dibromoterephthalic acid serves as an intermediate in pharmaceutical and agrochemical research. The bromine atoms enable cross-coupling reactions, such as Suzuki-Miyaura and Heck reactions, facilitating the construction of complex molecules with potential biological activity. Its derivatives have been explored for their roles in designing drugs and agrochemicals with improved efficacy and selectivity. Furthermore, the compound has applications in materials chemistry, contributing to the development of optoelectronic materials, dyes, and coatings. The electron-donating and withdrawing properties of its functional groups make it a valuable component in tailoring the electronic properties of advanced materials. The discovery and utilization of 2,5-dibromoterephthalic acid underscore its importance in modern chemistry. Its versatile reactivity and role in creating innovative materials highlight its significance in advancing multiple scientific and industrial fields. References 2023. Synthesis, crystal structure and antimicrobial activities of two silver(I) complexes based on bis(pyrazole) and carboxylic acid co-ligands. Transition Metal Chemistry, 48(6). DOI: 10.1007/s11243-023-00556-5 2023. A Novel Polythiophene-Conjugated Polymer Photocatalyst Under Visible Light for Killing Multidrug-Resistant Bacteria. Catalysis Letters, 153(8). DOI: 10.1007/s10562-023-04409-9 2023. Tuning the Dimensions of Coordination Polymers through Halogen Substituent Groups of Ligands. Russian Journal of General Chemistry, 93(8). DOI: 10.1134/s107036322308025x 2023. Click postsynthesis of microporous organic network@silica composites for reversed-phase/hydrophilic interaction mixed-mode chromatography. Analytical and Bioanalytical Chemistry, 415(14). DOI: 10.1007/s00216-023-04680-0 2008. 2,5-Dibromo-terephthalic acid dihydrate. Acta Crystallographica. Section E, Structure Reports Online, 64(Pt 9). DOI: 10.1107/s1600536808027268 |
| Market Analysis Reports |