Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Xi'an Union Pharmpro Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.unipharmpro.com | |||
 | +86 (29) 8886-5676 | |||
 | +86 (29) 8886-3915 | |||
 | info@unipharmpro.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Haimen RuiYi Medical Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ruiyitech.cn | |||
 | +86 (21) 5471-1706 | |||
 | +86 (21) 5471-1707 | |||
 | sales@ruiyitech.com zyj3625@msn.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| The Dow Chemical Company | USA | |||
|---|---|---|---|---|
 | www.dow.com | |||
 | +1 (989) 633-1706 | |||
 | fmdcigs@dow.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Langfang GreatAp Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.grechembld.com | |||
 | +86 (316) 209-8955 +86 18630626679 | |||
 | +86 (316) 209-8956 | |||
 | sales@grechembld.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Earlier HK Company Limited | China | |||
|---|---|---|---|---|
 | www.earlierchem.com | |||
 | +86 (10) 6142-6769 +86 18616907430 | |||
 | +86 (10) 6142-6769 | |||
 | info@earlierchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Kylin Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kylinchemicals.com | |||
 | +86 (519) 6878-1297 +86 15995066920 | |||
 | +86 (519) 8158-3863 | |||
 | info@kylinchemicals.com | |||
 | QQ Chat | |||
| Chemical distributor since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Saian Chemical (shanghai) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.saianchem.com | |||
 | +86 18633110822 | |||
 | teresa.liu@saianchem.com | |||
 | QQ Chat | |||
 | WeChat: 18633110822 | |||
| Chemical distributor since 2018 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Shanghai 3W Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.3wchem.com | |||
 | +86 (21) 6380-4615 5355-0305 | |||
 | +86 (21) 6381-4984 / 5371-2862 | |||
 | sale@3wchem.com | |||
| Chemical manufacturer since 1993 | ||||
| Qidong Sanli Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qdslchem.com | |||
 | +86 (513) 8391-7000 8388-5888 8391-0999 | |||
 | +86 (513) 8391-0666 | |||
 | slhg@hi2000.com | |||
| Chemical manufacturer | ||||
| Materion | USA | |||
|---|---|---|---|---|
 | www.materion.com | |||
 | +1 (216) 486-4200 | |||
 | +1 (414) 289-9805 | |||
 | info@materion.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Boride, borate and perborate |
|---|---|
| Name | Potassium borohydride |
| Synonyms | Potassium tetrahydroborate |
| Molecular Structure | 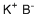 |
| Molecular Formula | KBH4 |
| Molecular Weight | 53.93 |
| CAS Registry Number | 13762-51-1 |
| EC Number | 237-360-5 |
| SMILES | [B-].[K+] |
| Density | 1.17 g/mL (Expl.) |
|---|---|
| Melting point | 400 °C (Decomposes) (Expl.) |
| Solubility | Soluble in water (19g/100g ). methanol (3.9g/100g ). 4:1 water:methanol mixture (13g/100g ) (Expl.) |
| Refraction index | 1.494 (Expl.) |
| Hazard Symbols |    GHS02;GHS05;GHS06 Danger Details GHS02;GHS05;GHS06 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H260-H301-H301-H311-H314-H318-H330 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P223-P231+P232-P260-P262-P264-P264+P265-P270-P271-P280-P284-P301+P316-P301+P330+P331-P302+P335+P334-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P320-P321-P330-P361+P364-P363-P370+P378-P402+P404-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1870 | ||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Potassium borohydride, with the chemical formula KBH4, is an inorganic compound consisting of potassium cations (K+) and tetrahydroborate anions (BH4−). It is a white, crystalline solid that is stable in dry air but reacts slowly with water and more rapidly with acids. It is a known reducing agent, particularly valued for its selective reactivity and stability compared to other borohydride salts such as sodium borohydride. The compound has been studied extensively for its chemical behavior and practical applications in laboratory and industrial settings. Potassium borohydride was first synthesized and characterized in the mid-20th century, following the successful development of related borohydrides like lithium borohydride and sodium borohydride. It can be prepared by the reaction of boron trifluoride etherate with potassium hydride or by metathesis reactions involving sodium borohydride and potassium-containing salts under controlled conditions. The resulting solid is typically isolated under inert atmosphere conditions to prevent hydrolysis and degradation. As a reducing agent, potassium borohydride is used in both organic and inorganic chemistry. It can reduce aldehydes, ketones, acid chlorides, and certain imines, though it is generally less reactive than sodium borohydride. Its relative mildness and lower solubility in protic solvents like water or alcohols can be advantageous in situations where controlled reactivity is required. This selective reducing power has made potassium borohydride useful in fine chemical synthesis and laboratory-scale preparations where precise control over reaction conditions is essential. In aqueous solutions, potassium borohydride exhibits greater hydrolytic stability than sodium borohydride, making it suitable for applications in which gradual hydrogen release is desired. The compound reacts with water to generate hydrogen gas, albeit more slowly than sodium borohydride. This property has been explored for potential hydrogen generation and storage systems, though large-scale adoption has been limited due to cost and availability. Nevertheless, its controlled hydrogen evolution has been beneficial in certain chemical reduction processes where gaseous hydrogen can be used in situ. In metallurgy and electroplating, potassium borohydride has been used as a reducing agent for metal ions in solution, allowing the deposition of metals such as nickel and cobalt onto substrates. It plays a role in electroless plating processes, where its reducing power initiates and sustains metal deposition without the need for an external electrical current. This application has been especially valuable in coating non-conductive surfaces and manufacturing components with uniform metallic finishes. The compound has also found utility in the stabilization of metal nanoparticles. In colloidal chemistry, potassium borohydride is used to reduce metal salts in solution, forming finely dispersed metal particles that are stabilized by suitable capping agents. These nanoparticles have potential applications in catalysis, electronics, and materials science, where controlled particle size and distribution are crucial. Potassium borohydride has been examined for use in pharmaceuticals and biochemical studies, primarily as a reagent for the reduction of carbonyl-containing compounds in drug intermediates. Its low toxicity compared to some other hydride reagents, combined with its ease of handling in dry form, makes it suitable for laboratory use. However, its commercial use in pharmaceutical production is less widespread due to the availability of more cost-effective alternatives. From a safety perspective, potassium borohydride must be handled with care. Although it is stable in dry air, it reacts with moisture and acids to release hydrogen gas, which is flammable and potentially explosive in confined environments. Proper storage in tightly sealed containers under dry conditions is required. Direct contact with water or humid air should be avoided to minimize risk. In the event of decomposition, toxic boron-containing compounds may be released, necessitating the use of appropriate ventilation and protective equipment. In summary, potassium borohydride is a well-characterized, stable, and versatile reducing agent with numerous applications in chemical synthesis, metallurgical processes, hydrogen generation, and materials science. Its unique balance of stability and reactivity distinguishes it from other borohydride salts and underpins its continued relevance in both academic research and applied chemistry. References 1959. Quantitative Determination of Flavanones in Citrus Bioflavonoids by Potassium Borohydride Reduction. Journal of the American Pharmaceutical Association, 48(12). DOI: 10.1002/jps.3030481214 2003. New Direct 11B NMR-Based Analysis of Organoboranes through Their Potassium Borohydrides. The Journal of Organic Chemistry, 68(11). DOI: 10.1021/jo020736+ 2011. Reduction of Nitroarenes to Azoxybenzenes by Potassium Borohydride in Water. Molecules, 16(5). DOI: 10.3390/molecules16053563 |
| Market Analysis Reports |