Online Database of Chemicals from Around the World
| Chengdu Hongsheng Sci-Tech Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cdchemical.com | |||
 | +86 (28) 3769-0186 +86 13568974161 | |||
 | +86 (28) 8587-6205 | |||
 | cdchemical@163.com cdchemi@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Sichuan Weibo Science and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.scweibo.com | |||
 | +86 (28) 8757-8473 +86 13880582998 | |||
 | +86 (28) 8757-8473 | |||
 | dudes@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Haimen RuiYi Medical Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ruiyitech.cn | |||
 | +86 (21) 5471-1706 | |||
 | +86 (21) 5471-1707 | |||
 | sales@ruiyitech.com zyj3625@msn.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Nanjing SiSiB Silicones Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sisib.com | |||
 | +86 (25) 5859-9930 5859-9931 5859-9932 +86 13913891929 | |||
 | +86 (25) 5859-9935 | |||
 | sales@sisib.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Infinity Scientific | China | |||
|---|---|---|---|---|
 | www.infsci.com | |||
 | +86 400-106-2016 +86 18500163824 | |||
 | biz2@infsci.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Finetech Industry Limited | China | |||
|---|---|---|---|---|
 | www.finetechchem.com | |||
 | +86 (27) 8746-5837 +86 18971612321 | |||
 | +86 (27) 8777-2287 | |||
 | sales@finetechnology-ind.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Mahidhara Chemicals Private Limited | India | |||
|---|---|---|---|---|
 | www.mahidhara.com | |||
 | +91 8455 240445 | |||
 | mahidharamarketing1@gmail.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical reagent >> Silane reagent |
|---|---|
| Name | Potassium bis(trimethylsilyl)amide |
| Synonyms | Potassiumhexamethyldisilazane |
| Molecular Structure | 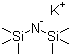 |
| Molecular Formula | C6H18KNSi2 |
| Molecular Weight | 199.48 |
| CAS Registry Number | 40949-94-8 |
| EC Number | 609-880-6 |
| SMILES | C[Si](C)(C)[N-][Si](C)(C)C.[K+] |
| Density | 0.881 g/mL (Expl.) |
|---|---|
| Index of Refraction | 1.492 (Expl.) |
| Boiling point | 66 °C °C (Expl.) |
| Flash point | -14 °C °C (Expl.) |
| Hazard Symbols |  GHS05 Danger Details GHS05 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H318 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P264-P264+P265-P280-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2924 | ||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Potassium bis(trimethylsilyl)amide (KBMDS) is an important potassium-based organometallic reagent that has found widespread use in organic synthesis due to its strong basicity and ability to form reactive intermediates. Like its lithium counterpart, lithium bis(trimethylsilyl)amide (LiHMDS), KBMDS contains the bis(trimethylsilyl)amide anion, but it features potassium instead of lithium as the cation. This difference in cation results in differences in reactivity and solubility, which can be exploited in various synthetic applications. KBMDS was developed as part of the ongoing effort to create more versatile and efficient organometallic reagents for use in organic chemistry. While lithium reagents have been central to synthetic strategies for decades, the development of potassium analogs like KBMDS has provided chemists with additional options for reactions requiring strong bases. The introduction of potassium bis(trimethylsilyl)amide emerged as a way to overcome some limitations associated with lithium-based reagents, including differences in solubility and reactivity, offering an alternative for specific reactions. KBMDS is typically synthesized by reacting potassium metal with bis(trimethylsilyl)amine under anhydrous conditions, in solvents like tetrahydrofuran (THF) or diethyl ether. The reagent is usually used in its dry, powdery form, as moisture and air can lead to hydrolysis, which renders the reagent inactive. Its high solubility in nonpolar solvents, such as THF, allows it to be used effectively in a wide range of reactions involving organic substrates. One of the primary applications of KBMDS lies in its use as a strong base in organic synthesis. Due to its high basicity, KBMDS can deprotonate a variety of compounds, particularly weak acids. This property is exploited in the formation of reactive intermediates like enolates, which can be used in nucleophilic addition reactions. KBMDS is especially useful for generating enolates from carbonyl compounds, such as ketones and aldehydes, which are crucial intermediates in many organic transformations, including aldol reactions, Michael additions, and other carbon-carbon bond-forming reactions. Additionally, KBMDS is employed in the synthesis of organometallic intermediates, such as organozinc, organoboron, and organosilicon compounds. These intermediates are key players in a range of cross-coupling reactions, such as the Suzuki, Negishi, and Kumada couplings, which are frequently used in the synthesis of complex organic molecules. In particular, the ability of KBMDS to generate organozinc compounds is valuable in these reactions, as these reagents can participate in highly selective carbon-carbon bond formations, enabling the efficient synthesis of natural products, pharmaceuticals, and agrochemicals. KBMDS is also utilized in reactions that require strong bases to induce deprotonation or to initiate nucleophilic substitution reactions. For example, it is used to promote the dehydrohalogenation of alkyl halides, a step commonly required in the preparation of alkenes, and is effective in the synthesis of other organic functionalities, such as alkynes, amides, and lactams. In addition to its role in synthetic organic chemistry, KBMDS has applications in materials science, particularly in the preparation of silane-based polymers and coatings. As a source of the bis(trimethylsilyl)amide group, KBMDS is used in the synthesis of siloxanes, which have applications in industries such as electronics, adhesives, and coatings. Its reactivity allows for the development of functionalized silanes and polysiloxanes with specific properties tailored to various technological applications. The strong basicity and versatility of KBMDS make it a valuable reagent in both academic research and industrial applications. However, due to its highly reactive nature, it must be handled with caution. Proper storage under dry, inert conditions and the use of anhydrous solvents are critical to maintaining the stability and reactivity of KBMDS. References 2024. Nucleophilic trifluoromethylation with CF3H/LiHMDS: probing the nucleophilic reactivity of LiCF3 species. Science China Chemistry, 67(10). DOI: 10.1007/s11426-024-2289-7 2024. Autonomous execution of highly reactive chemical transformations in the Schlenkputer. Nature Chemical Engineering, 1(2). DOI: 10.1038/s44286-023-00024-y 2023. Catalytic production of ammonia from dinitrogen employing molybdenum complexes bearing N-heterocyclic carbene-based PCP-type pincer ligands. Nature Synthesis, 2(6). DOI: 10.1038/s44160-023-00292-9 |
| Market Analysis Reports |