Hydrazinium bromide, with the chemical formula N2H8Br, is an inorganic compound that has garnered attention due to its unique chemical properties and potential applications. The discovery of hydrazinium bromide is linked to the broader exploration of hydrazine derivatives, which began in the early 20th century. Hydrazine itself was first synthesized in 1887 by the German chemist Hermann Emil Fischer. Over the years, various derivatives of hydrazine, including hydrazinium bromide, were developed to explore their applications in diverse fields.
Hydrazinium bromide is primarily recognized for its role as a reducing agent and as a precursor in the synthesis of other chemical compounds. Its structure features a hydrazinium cation (N2H8^+) and a bromide anion (Br^-), which contribute to its reactivity. The compound is often utilized in organic synthesis, particularly in the formation of nitrogen-containing compounds. This characteristic makes it valuable in the production of pharmaceuticals, agrochemicals, and fine chemicals.
One of the notable applications of hydrazinium bromide is in the field of organic chemistry as a reagent for the reduction of various functional groups. For instance, it has been employed to reduce aromatic nitro compounds to amines, facilitating the synthesis of important intermediates used in pharmaceuticals. Additionally, hydrazinium bromide can be utilized in the preparation of hydrazone derivatives, which are significant in medicinal chemistry due to their biological activities.
Moreover, hydrazinium bromide has been explored in the context of polymer chemistry. It can act as a chain transfer agent in radical polymerization processes, influencing the molecular weight and distribution of polymer products. This property allows for the tailored design of polymer materials with specific characteristics, which can be advantageous in various industrial applications.
In the realm of materials science, hydrazinium bromide has been investigated for its potential use in the preparation of energetic materials. Its nitrogen-rich structure provides a pathway for the development of new propellants and explosives. Researchers have explored its properties to assess its stability and performance, contributing to advancements in the field of energetic materials.
Despite its various applications, handling hydrazinium bromide requires caution due to its potential toxicity and reactivity. Safety measures should be observed when working with this compound, particularly in laboratory settings, to mitigate any risks associated with its use.
In summary, hydrazinium bromide is a versatile compound with origins in the early exploration of hydrazine derivatives. Its applications in organic synthesis, polymer chemistry, and materials science highlight its significance in various chemical processes. Ongoing research continues to unveil the potential of hydrazinium bromide in developing new materials and chemical intermediates, contributing to advancements in multiple fields.
References
1955. Protection of workers against dermatitis on exposure to hydrazine hydrobromide soldering flux. International Archives of Occupational and Environmental Health.
DOI: 10.1007/bf00316713
2000. The Electronics Industry. Handbook of Occupational Dermatology.
DOI: 10.1007/978-3-662-07677-4_82
1967. Synthesis of some o-carborane derivatives. Chemistry of Heterocyclic Compounds.
DOI: 10.1007/bf00945397
|













































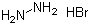



 GHS06;GHS07;GHS08;GHS09 Danger Details
GHS06;GHS07;GHS08;GHS09 Danger Details