Online Database of Chemicals from Around the World
| Xiamen Pioneer Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xm-pioneer.com | |||
 | +86 (592) 392-2797 | |||
 | +86 (592) 377-6077 | |||
 | xmpt@xm-pioneer.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Dishman Pharmaceuticals & Chemicals Limited | USA | |||
|---|---|---|---|---|
 | www.dishmangroup.com | |||
 | +1 (732) 560-4300 | |||
 | +1 (732) 560-4343 | |||
 | bhaveshoza@dishmangroup.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Guizhou Jianye Yongrong Technology Co., Ltd. (Changzhou Huadong Chemical Research Institute) | China | |||
|---|---|---|---|---|
 | www.huadongchem.com | |||
 | +86 +13851563211 | |||
 | +86 (519) 8261-6386 | |||
 | sales@huadongchem.com | |||
 | WeChat: +13851563211 | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Shanghai Cainorise Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cainorise.com.cn | |||
 | +86 (21) 3408-0216 2950-8524 +86 15921434758 | |||
 | sales@cainorise.com.cn | |||
| Chemical manufacturer | ||||
| Pressure Chemical Company | USA | |||
|---|---|---|---|---|
 | www.presschem.com | |||
 | +1 (800) 722-5247 (412) 682-5882 | |||
 | +1 (412) 682-5864 | |||
 | contact_us@presschem.com | |||
| Chemical manufacturer since 1964 | ||||
| Classification | Chemical reagent >> Organic reagent >> Amine salt (ammonium salt) |
|---|---|
| Name | Phenyltrimethylammonium chloride |
| Synonyms | N,N,N-Trimethylanilinium chloride; Trimethylphenylammonium chloride |
| Molecular Structure | 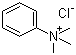 |
| Molecular Formula | C9H14N.Cl |
| Molecular Weight | 171.67 |
| CAS Registry Number | 138-24-9 |
| EC Number | 205-319-0 |
| SMILES | C[N+](C)(C)C1=CC=CC=C1.[Cl-] |
| Melting point | 246-248 °C (dec.) (Expl.) |
|---|---|
| Water solubility | soluble |
| Hazard Symbols |  GHS06 Danger Details GHS06 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H311 Details | ||||||||||||||||||||||||||||
| Safety Statements | P262-P264-P270-P280-P301+P316-P302+P352-P316-P321-P330-P361+P364-P405- Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Phenyltrimethylammonium chloride (C6H5N+(CH3)3Cl) is a quaternary ammonium salt that consists of a phenyl group attached to a trimethylammonium cation (C6H5N+(CH3)3) and a chloride anion (Cl−). It is synthesized by reacting trimethylamine with benzyl chloride or phenylmethyl chloride in an appropriate solvent. Phenyltrimethylammonium chloride is commonly used as a phase transfer catalyst in organic synthesis. A phase transfer catalyst facilitates the transfer of reactants between two immiscible phases, often between water and an organic solvent. The compound’s quaternary ammonium structure allows it to solvate anionic species and transfer them into organic solvents, where they would otherwise be insoluble. This ability to assist in the movement of ions between phases is particularly useful in reactions involving anions, where it enhances the reactivity of certain nucleophiles in organic reactions. Its primary application is in organic synthesis, particularly in nucleophilic substitution reactions, where it aids in the formation of various organic compounds. For instance, phenyltrimethylammonium chloride can be used to catalyze the alkylation of phenols, the preparation of alkyl and aryl derivatives, and various other transformations involving halides and other reactive anionic species. Additionally, it has been utilized in reactions where ionic species need to be transferred between aqueous and organic phases to facilitate efficient reactions. The compound has also found use in research as a reagent for various synthetic applications due to its phase transfer capabilities. In some cases, it is employed to aid in the solubilization of water-soluble anions in nonpolar solvents or to promote the synthesis of other quaternary ammonium salts. In summary, phenyltrimethylammonium chloride is an important chemical used in organic synthesis, primarily as a phase transfer catalyst. It plays a critical role in facilitating reactions that require the transfer of anionic species between immiscible phases, thereby enhancing the efficiency of various chemical transformations. References 2024. Self-powered and charge-transport-layer-free photodetectors based on chloride treated perovskites for weak light detection. Science China Materials, 67(6). DOI: 10.1007/s40843-023-2832-y 2024. Phase stabilization of cesium lead iodide perovskites for use in efficient optoelectronic devices. NPG Asia Materials, 16(1). DOI: 10.1038/s41427-024-00540-0 2024. Weakly Polarized Organic Cation-Modified Hydrated Vanadium Oxides for High-Energy Efficiency Aqueous Zinc-Ion Batteries. Nano-Micro Letters, 16(1). DOI: 10.1007/s40820-024-01339-y |
| Market Analysis Reports |