Online Database of Chemicals from Around the World
| Jinan Orgachem Technology and Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.orgachem.com | |||
 | +86 (531) 8268-7809 8268-7823 8268-7819 | |||
 | +86 (531) 8268-7807 | |||
 | dzgdshyq@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Shanghai Jinhuan Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jinhchem.com | |||
 | +86 (21) 6142-0008 | |||
 | +86 (21) 5662-4506 | |||
 | chenhui79820@hotmail.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhejiang Ouhua Chemical Imp. & Exp. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ouhuachem.com | |||
 | +86 (576) 8882-6717 | |||
 | +86 (576) 8882-8330 | |||
 | ouhuachem@gmail.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2009 | ||||
| Arshine Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.arshine.com.cn | |||
 | +86 (755) 3333-0822 | |||
 | +86 (755) 8287-9323 | |||
 | marketing@arshine.com.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Winchem Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-chemical.com | |||
 | +86 (574) 8385-1061 | |||
 | +86 (574) 8708-3209 | |||
 | info@win-chemical.com hhp13@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Turtle Pharma Private Limited | India | |||
|---|---|---|---|---|
 | www.turtlepharma.com | |||
 | +91 9850998504 | |||
 | info@turtlepharma.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Dezhou Deyao Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dzzy.cn | |||
 | +86 (29) 8831-8318 ex 1682 8815-2420 | |||
 | +86 (29) 8815-2414 / 8815-2421 | |||
 | webmaster@dzzy.cn | |||
| Chemical manufacturer | ||||
| Classification | API >> Synthetic anti-infective drugs >> Quinolone |
|---|---|
| Name | Levofloxacin hydrate |
| Synonyms | (S)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido(1,2,3-de)-1,4-benzoxazine-6-carboxylic acid hydrate (2:1) |
| Molecular Structure | 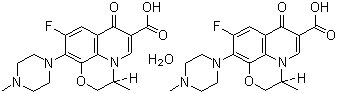 |
| Molecular Formula | 2(C18H20FN3O4).H2O |
| Molecular Weight | 740.75 |
| CAS Registry Number | 138199-71-0 |
| EC Number | 604-067-2 |
| SMILES | C[C@H]1COC2=C3N1C=C(C(=O)C3=CC(=C2N4CCN(CC4)C)F)C(=O)O.C[C@H]1COC2=C3N1C=C(C(=O)C3=CC(=C2N4CCN(CC4)C)F)C(=O)O.O |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H317-H334-H361-H362 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P233-P260-P261-P263-P264-P270-P271-P272-P280-P284-P301+P317-P302+P352-P304+P340-P318-P321-P330-P333+P317-P342+P316-P362+P364-P403-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Levofloxacin hydrate is a potent antibiotic belonging to the fluoroquinolone class, renowned for its broad-spectrum antibacterial activity. Developed as a third-generation fluoroquinolone, levofloxacin hydrate represents an important advancement in the treatment of bacterial infections, with a range of applications across various medical fields. The discovery of levofloxacin began with the development of earlier fluoroquinolones, such as ciprofloxacin and norfloxacin, which demonstrated significant antibacterial efficacy. Levofloxacin is the levorotatory isomer of ofloxacin, a second-generation fluoroquinolone. The transition to levofloxacin involved refining the molecular structure to enhance its potency and extend its spectrum of activity. It was introduced in the 1990s as a more effective option against a wider range of pathogens. Levofloxacin works by inhibiting bacterial DNA gyrase and topoisomerase IV—enzymes crucial for DNA replication and transcription. This inhibition effectively prevents bacterial cell division, leading to the death of the bacteria. Its broad-spectrum activity covers a variety of Gram-positive and Gram-negative bacteria, including strains resistant to other antibiotics. One of the primary applications of levofloxacin hydrate is in the treatment of respiratory infections. It is commonly prescribed for conditions such as community-acquired pneumonia, chronic bronchitis, and sinusitis. Its ability to target pathogens like *Streptococcus pneumoniae* and *Haemophilus influenzae* makes it highly effective in managing these infections. In addition to respiratory infections, levofloxacin hydrate is used to treat urinary tract infections (UTIs), including complicated UTIs and pyelonephritis. It is also employed in treating skin infections, such as cellulitis and infected wounds, and in some cases, it is used for certain types of intra-abdominal infections. Levofloxacin hydrate is available in both oral and intravenous formulations, providing flexibility in treatment administration. The oral tablets allow for convenient outpatient management, while the intravenous form is useful in hospital settings for severe infections or when oral administration is not feasible. One of the advantages of levofloxacin hydrate is its favorable pharmacokinetic profile. It is well-absorbed when taken orally, with good tissue penetration, ensuring effective drug levels at infection sites. The drug's long half-life allows for once-daily dosing, which can improve patient adherence and simplify treatment regimens. Despite its benefits, the use of levofloxacin hydrate must be managed carefully to minimize the risk of developing antibiotic resistance. Overuse or misuse of fluoroquinolones can lead to resistance, which poses a significant challenge in treating bacterial infections. Guidelines recommend using levofloxacin for infections where its benefits outweigh the risks and avoiding it for less severe infections when other antibiotics might be effective. Levofloxacin hydrate can also have side effects, including gastrointestinal disturbances, central nervous system effects such as dizziness or headaches, and in rare cases, tendon damage or rupture. Patients are advised to report any severe or unusual symptoms to their healthcare provider. In summary, levofloxacin hydrate is a valuable antibiotic in the fluoroquinolone class with a broad range of applications. Its development has enhanced the ability to treat various bacterial infections effectively, particularly those resistant to older antibiotics. As with all antibiotics, prudent use is essential to maintain its effectiveness and mitigate the risks of resistance. References 1991. Three-dimensional structure-activity relationships and receptor mapping of N1-substituents of quinolone antibacterials. Journal of Medicinal Chemistry. DOI: 10.1021/jm00105a020 2003. Antibiotic Therapy for Ambulatory Patients With Community-Acquired Pneumonia in an Emergency Department Setting. Archives of Internal Medicine. DOI: 10.1001/archinte.163.7.797 2025. Gastro retentive floating drug delivery system of levofloxacin based on Aloe vera hydrogel: In vitro and in vivo assays. International Journal of Biological Macromolecules. DOI: 10.1016/j.ijbiomac.2024.138156 |
| Market Analysis Reports |