Online Database of Chemicals from Around the World
| Pure Chemistry Scientific Inc | USA | |||
|---|---|---|---|---|
 | www.purechems.com | |||
 | +1 (888) 588-9418 | |||
 | +1 (617) 206-9595 | |||
 | sales@purechems.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| HetCat - Heterocycles & Catalysts | Switzerland | |||
|---|---|---|---|---|
 | www.hetcat.com | |||
 | +41 (61) 363-9868 | |||
 | +41 (61) 363-9869 | |||
 | fallahpour@hetcat.com | |||
| Chemical manufacturer | ||||
| MCAT GmbH | Germany | |||
|---|---|---|---|---|
 | www.mcat.de | |||
 | +49 (7531) 939-097 | |||
 | +49 (7531) 939-098 | |||
 | info@mcat.de | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Classification | Chemical reagent >> Organic reagent >> Borate |
|---|---|
| Name | Bis(catecholato)diborane |
| Synonyms | 2,2'-Bis-1,3,2-benzodioxaborole |
| Molecular Structure | 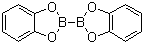 |
| Molecular Formula | C12H8B2O4 |
| Molecular Weight | 237.81 |
| CAS Registry Number | 13826-27-2 |
| EC Number | 691-473-8 |
| SMILES | B1(OC2=CC=CC=C2O1)B3OC4=CC=CC=C4O3 |
| Melting point | 189-196 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Bis(catecholato)diborane is a notable compound in the field of organoboron chemistry, recognized for its unique structure and diverse applications. This compound, with the chemical formula B2(C6H4O2)2, consists of a diborane core bonded to two catecholato (1,2-dihydroxybenzene) ligands. Its structure represents an interesting example of how boron chemistry can be combined with aromatic systems to create functional materials. The discovery of bis(catecholato)diborane emerged from research focused on expanding the range of organoboron compounds and exploring their potential applications. Diborane, a fundamental compound in boron chemistry, has been extensively studied for its reactivity and ability to form complex structures with various ligands. The introduction of catecholato ligands to the diborane core enhances the compound's stability and introduces new chemical properties. The synthesis of bis(catecholato)diborane typically involves the reaction of diborane with catechol or its derivatives under controlled conditions. This process requires careful handling due to the reactivity of diborane. The formation of bis(catecholato)diborane is achieved by coordinating catecholato ligands to the diborane center, resulting in a stable complex that retains the distinctive features of both components. One of the key applications of bis(catecholato)diborane is in the field of materials science. The compound's structure allows it to act as a precursor for the synthesis of boron-containing materials with specific properties. For instance, bis(catecholato)diborane can be used to create advanced ceramics, boron-doped semiconductors, and other materials with tailored electronic or optical properties. These materials are valuable in various industrial applications, including electronics, energy storage, and catalysis. In addition to its role in materials science, bis(catecholato)diborane has potential applications in catalysis. The presence of catecholato ligands can influence the compound's ability to act as a catalyst or catalyst support in chemical reactions. Researchers have explored its use in various catalytic processes, including those related to organic synthesis and environmental remediation. The catecholato ligands can participate in coordination and bonding interactions, enhancing the compound's catalytic activity. The compound is also of interest in the study of boron chemistry and the development of new boron-based reagents. Bis(catecholato)diborane can be used as a building block for the synthesis of more complex boron-containing compounds. Its ability to form stable complexes with other molecules makes it a valuable tool for researchers investigating the chemistry of boron and its interactions with different ligands. The reactivity of bis(catecholato)diborane is influenced by the presence of catecholato ligands, which can engage in hydrogen bonding and other interactions. This property makes it useful for studying the fundamental interactions of boron compounds with other substances. Researchers have used bis(catecholato)diborane to explore how catecholato ligands affect the properties and behavior of boron hydrides. The synthesis of bis(catecholato)diborane involves precise control of reaction conditions, including the concentration of reactants, temperature, and reaction time. Typically, the compound is obtained as a crystalline solid, which can be characterized using techniques such as X-ray crystallography and spectroscopy. Overall, bis(catecholato)diborane is a valuable compound in organoboron chemistry with diverse applications in materials science, catalysis, and chemical synthesis. Its unique structure and properties provide opportunities for developing new materials and exploring the chemistry of boron. Continued research into this compound is likely to reveal further applications and enhance our understanding of boron-containing compounds. References 2010. Perceptible Influence of Pd and Pt Heterocyclic Carbene-Pyridyl Complexes in Catalytic Diboration of Cyclic Alkenes. Chemistry - An Asian Journal. DOI: 10.1002/asia.200900580 2019. Light-Induced Reactions. Science of Synthesis. DOI: https://science-of-synthesis.thieme.com/app/text/?id=SD-230-00168 2021. Tetrahydroxydiboron-Initiated Atom-Transfer Radical Cyclization. Synthesis. DOI: 10.1055/a-1485-4956 |
| Market Analysis Reports |