Online Database of Chemicals from Around the World
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhengzhou Changkuan Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.changkuantech.com | |||
 | +86 (371) 6376-9919 +86 18530983798 | |||
 | +86 (371) 6360-3986 | |||
 | sales5@changkuantech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Amide |
|---|---|
| Name | (1R,2R)-(+)-N,N'-Bis(2-diphenylphosphinobenzoyl)-1,2-diaminocyclohexane |
| Synonyms | (R,R)-DACH-phenyl Trost Ligand |
| Molecular Structure | 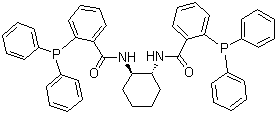 |
| Molecular Formula | C44H40N2O2P2 |
| Molecular Weight | 690.75 |
| CAS Registry Number | 138517-61-0 |
| EC Number | 811-290-0 |
| SMILES | C1CC[C@H]([C@@H](C1)NC(=O)C2=CC=CC=C2P(C3=CC=CC=C3)C4=CC=CC=C4)NC(=O)C5=CC=CC=C5P(C6=CC=CC=C6)C7=CC=CC=C7 |
| Melting point | 136-142 °C |
|---|---|
| alpha | 130 |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315 Details |
| Safety Statements | P264-P280-P302+P352-P321-P332+P317-P362+P364 Details |
| SDS | Available |
|
(1R,2R)-(+)-N,N'-Bis(2-diphenylphosphinobenzoyl)-1,2-diaminocyclohexane is a noteworthy compound in the field of organometallic chemistry, particularly for its role as a chiral ligand in asymmetric synthesis. This compound combines a chiral diamine backbone with two diphenylphosphinobenzoyl groups, resulting in a ligand with remarkable properties for catalytic applications. The compound was introduced as part of ongoing efforts to develop new chiral ligands that can facilitate asymmetric transformations with high selectivity. Asymmetric synthesis is a crucial area in chemistry, enabling the production of chiral molecules with high enantiomeric purity. The design of effective chiral ligands plays a vital role in these reactions, as they can significantly influence the stereoselectivity and efficiency of the catalysis. The synthesis of (1R,2R)-(+)-N,N'-Bis(2-diphenylphosphinobenzoyl)-1,2-diaminocyclohexane involves the attachment of two diphenylphosphinobenzoyl groups to a chiral 1,2-diaminocyclohexane framework. The cyclohexane ring in this ligand provides a rigid, chiral environment that stabilizes the coordination of metal centers, while the diphenylphosphinobenzoyl groups enhance the electronic and steric properties of the ligand. One of the significant applications of this compound is in asymmetric catalysis. In particular, it has been used in asymmetric hydrogenation and other reactions where the generation of chiral products is desired. The combination of the chiral diamine and the diphenylphosphinobenzoyl groups creates an environment that can effectively discriminate between different enantiomers, leading to high enantioselectivity in catalytic processes. The effectiveness of (1R,2R)-(+)-N,N'-Bis(2-diphenylphosphinobenzoyl)-1,2-diaminocyclohexane as a ligand has been demonstrated in various catalytic reactions. For instance, its application in asymmetric hydrogenation has shown that it can provide excellent enantioselectivity, making it a valuable tool for synthesizing chiral molecules in the pharmaceutical and fine chemicals industries. In addition to its role in asymmetric synthesis, this compound has also contributed to the development of new methodologies in organometallic chemistry. By exploring different combinations of chiral ligands and metal catalysts, researchers have been able to improve reaction conditions, expand the scope of reactions, and achieve higher yields and selectivities. The discovery and application of (1R,2R)-(+)-N,N'-Bis(2-diphenylphosphinobenzoyl)-1,2-diaminocyclohexane highlight the importance of ligand design in catalytic chemistry. Its introduction has provided chemists with a powerful tool for asymmetric synthesis, demonstrating the ongoing progress in developing ligands that enhance the efficiency and selectivity of catalytic processes. References 2005. New Bisphosphine Ligands for Palladium-Catalyzed Desymmetrization of meso-Cyclopent-2-en-1,4-diol Biscarbamate. Synlett. DOI: 10.1055/s-2005-871963 2022. Palladium-Catalyzed Allylic Substitution. Science of Synthesis. DOI: https://science-of-synthesis.thieme.com/app/text/?id=SD-237-00128 2016. Enantioselective Construction of Quaternary Carbon Stereocenter via Palladium-Catalyzed Asymmetric Allylic Alkylation of Lactones. Synthesis. DOI: 10.1055/s-0035-1561574 |
| Market Analysis Reports |