Online Database of Chemicals from Around the World
| Chizhou Dongsheng Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dspharmchem.com | |||
 | +86 (576) 8429-2571 | |||
 | +86 (576) 8429-2560 | |||
 | ksz@dspharmchem.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Chemical reagent >> Organic reagent >> Imine, amidine |
|---|---|
| Name | 2-Amidinopyrimidine hydrochloride |
| Synonyms | pyrimidine-2-carboximidamide;hydrochloride |
| Molecular Structure | 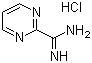 |
| Molecular Formula | C5H6N4.HCl |
| Molecular Weight | 158.59 |
| CAS Registry Number | 138588-40-6 |
| EC Number | 604-087-1 |
| SMILES | C1=CN=C(N=C1)C(=N)N.Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P312-P330-P332+P313-P337+P313-P362-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2-Amidinopyrimidine hydrochloride is an important heterocyclic compound with significant applications in the fields of pharmaceuticals and chemical research. The compound consists of a pyrimidine ring substituted with an amidino group (-C(=NH)NH2) at the second position, and it is typically isolated in the form of its hydrochloride salt to enhance its stability and solubility. The discovery of 2-amidinopyrimidine hydrochloride can be traced back to investigations into heterocyclic chemistry and nitrogen-containing bases. Pyrimidine derivatives have long been of interest due to their structural similarity to nucleic acid bases, such as cytosine and thymine, which play key roles in DNA and RNA. The amidine functional group further enhances the biological activity of these compounds by providing an additional site for hydrogen bonding and electrostatic interactions, which can be critical in enzyme inhibition and receptor binding. One of the major applications of 2-amidinopyrimidine hydrochloride is in medicinal chemistry. It has been explored as a core scaffold for the development of drugs targeting enzymes, particularly proteases. Due to its ability to mimic arginine-like residues, the compound can act as an inhibitor for certain enzymes involved in the regulation of coagulation, inflammation, and other physiological processes. The amidine group can form strong interactions with active site residues, enhancing the potency and selectivity of the inhibitors designed from this core structure. In addition to enzyme inhibition, 2-amidinopyrimidine hydrochloride has been studied for its antimicrobial properties. Pyrimidine derivatives with amidine functionalities have shown activity against a range of bacterial and parasitic infections, making them potential candidates for the development of new antibiotics or antiprotozoal drugs. These compounds are particularly valuable in addressing drug-resistant strains of bacteria, a growing concern in modern medicine. Furthermore, 2-amidinopyrimidine hydrochloride is utilized in synthetic organic chemistry as a building block for the construction of more complex molecules. Its reactivity, particularly due to the electron-donating nature of the amidine group, allows for diverse chemical transformations that can lead to a wide array of heterocyclic structures. This versatility has made it a valuable intermediate in the synthesis of various pharmaceutical agents and agrochemicals. Safety considerations are important when working with 2-amidinopyrimidine hydrochloride. Like many hydrochloride salts, it is hygroscopic and should be stored in tightly sealed containers to prevent moisture absorption. Additionally, appropriate handling procedures should be followed to avoid exposure, as the compound may cause irritation to the skin, eyes, or respiratory system. In summary, 2-amidinopyrimidine hydrochloride is a compound of interest due to its biological relevance and utility in drug development. Its ability to serve as an enzyme inhibitor, antimicrobial agent, and synthetic intermediate highlights its versatility and potential in both pharmaceutical and chemical research. References 2018. Synthesis of hybrid molecules containing pyrimidine and diterpene alkaloid lappaconitine fragments. Chemistry of Heterocyclic Compounds. DOI: 10.1007/s10593-019-02404-w 2019. Design, synthesis, cytotoxicity, and molecular modeling study of 2,4,6-trisubstituted pyrimidines with anthranilate ester moiety. Medicinal Chemistry Research. DOI: 10.1007/s00044-019-02314-8 |
| Market Analysis Reports |