Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Ooo Npo Termodinamika 3000 | Russia | |||
|---|---|---|---|---|
 | tdk3000.com | |||
 | +7 (903) 609-51-94 | |||
 | termodinamika3000@gmail.com | |||
 | WeChat: +7 (903) 609-51-94 | |||
 | WhatsApp:+7 (903) 609-51-94 | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Fujian Wolfa Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wolfabio.com | |||
 | +86 18050950397 | |||
 | jomin@wolfabio.com | |||
 | WhatsApp:+86 18359103607 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Gelest, Inc. | USA | |||
|---|---|---|---|---|
 | www.gelest.com | |||
 | +1 (215) 547-1015 | |||
 | +1 (215) 547-2484 | |||
 | info@gelest.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic scandium, tantalum, thallium, tungsten, antimony, lanthanum, lead, vanadium, molybdenum, chromium, ytterbium, etc. |
|---|---|
| Name | Molybdenum hexacarbonyl |
| Synonyms | carbon monoxide molybdenum |
| Molecular Structure | 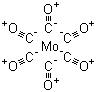 |
| Molecular Formula | C6MoO6 |
| Molecular Weight | 264.00 |
| CAS Registry Number | 13939-06-5 |
| EC Number | 237-713-3 |
| SMILES | [C-]#[O+].[C-]#[O+].[C-]#[O+].[C-]#[O+].[C-]#[O+].[C-]#[O+].[Mo] |
| Density | 1.96 |
|---|---|
| Melting point | 150 °C |
| Water solubility | insoluble |
| Hazard Symbols |  GHS06 Danger Details GHS06 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H300-H310-H330 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P260-P262-P264-P270-P271-P280-P284-P301+P316-P302+P352-P304+P340-P316-P320-P321-P330-P361+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 3466 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Molybdenum hexacarbonyl, with the chemical formula Mo(CO)₆, is a prominent organometallic compound known for its unique structure and diverse applications. The compound has played a significant role in advancing organometallic chemistry and has practical uses in various industrial and research settings. The discovery of molybdenum hexacarbonyl dates back to the early 20th century when scientists were exploring the chemistry of transition metal carbonyls. The synthesis of Mo(CO)₆ involves reacting molybdenum metal with carbon monoxide under specific conditions to form this stable hexacarbonyl complex. The compound features a structure in which a central molybdenum atom is coordinated by six carbon monoxide ligands in an octahedral arrangement. This configuration imparts notable electronic and steric properties to the complex. One of the primary applications of molybdenum hexacarbonyl is in catalysis. The compound is used as a catalyst in various chemical reactions, particularly in organic synthesis. It plays a crucial role in reactions such as hydrogenation and hydroformylation, where it facilitates the addition of hydrogen or carbonyl groups to organic substrates. Molybdenum hexacarbonyl's ability to form stable intermediates and participate in multiple reaction pathways makes it a valuable tool in the synthesis of complex organic molecules. In addition to its catalytic applications, molybdenum hexacarbonyl is used in the production of molybdenum-based materials. The compound can be decomposed to produce molybdenum metal or molybdenum carbides, which are used in high-performance materials and coatings. Molybdenum and its compounds are known for their high melting points and resistance to wear and corrosion, making them suitable for use in high-temperature environments and demanding industrial applications. Molybdenum hexacarbonyl also serves as a model compound for studying metal-carbonyl interactions and bonding. The well-defined structure of Mo(CO)₆ provides valuable insights into the nature of metal-carbonyl bonding and the electronic effects of carbonyl ligands on transition metals. Researchers use this compound to explore the principles of coordination chemistry and to develop new metal-carbonyl complexes with tailored properties. Handling molybdenum hexacarbonyl requires careful attention due to the toxicity of carbon monoxide. Proper safety measures, including working in well-ventilated areas and using appropriate protective equipment, are essential to prevent exposure to toxic fumes. Storage and handling procedures should follow established safety guidelines to ensure safe use in laboratory and industrial settings. Ongoing research continues to explore new applications and optimize the properties of molybdenum hexacarbonyl. Advances in organometallic chemistry and materials science are leading to the development of novel catalysts and materials based on this compound, with potential implications for various industrial and technological applications. In summary, molybdenum hexacarbonyl is an important organometallic compound with significant applications in catalysis, materials science, and the study of metal-carbonyl interactions. Its discovery and development have contributed to the field of chemistry, and continued research holds promise for discovering further uses and improvements. References 2024. Molybdenum Catalysts Supported on Porous Aromatic Frameworks in Epoxidation of Olefins. Petroleum Chemistry. DOI: 10.1134/s0965544124020208 2024. QTAIM analysis of the bonding in anionic group 6 carbonyl selenide clusters: [Se2M3(CO)10]2- (M=Cr, Mo, W). Journal of Molecular Modeling. DOI: 10.1007/s00894-024-06031-x 2024. Anchoring of boron halides BX (X: F, Cl, and Br) on transition metal (M: Cr, Mo, W) carbonyl complexes M(CO)5 (M: Cr, Mo, W): structure, bonding, and energy decomposition studies based on theoretical calculations. Structural Chemistry. DOI: 10.1007/s11224-024-02406-1 |
| Market Analysis Reports |