Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal halides and halides >> Metal fluorides and salts |
|---|---|
| Name | Molybdenum Hexafluoride |
| Synonyms | Molybdenum Fluoride; Molybdenum(6+) Fluoride; Molybdenum(Vi) Fluoride |
| Molecular Structure | 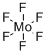 |
| Molecular Formula | F6Mo |
| Molecular Weight | 209.95 |
| CAS Registry Number | 7783-77-9 |
| EC Number | 232-026-5 |
| SMILES | F[Mo](F)(F)(F)(F)F |
| Density | 2.3 g/mL (Expl.) |
|---|---|
| Melting point | 17.5 °C (decomp.) (Expl.) |
| Boiling Point | 37 °C (decomp.) (Expl.) |
| Hazard Symbols |    GHS05;GHS06;GHS07 Danger Details GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H314-H318-H330 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P264-P264+P265-P270-P271-P280-P284-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P320-P321-P330-P362+P364-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
Molybdenum hexafluoride (MoF6) is a chemical compound consisting of a molybdenum atom surrounded by six fluorine atoms. It is classified as a hexafluoride, similar to other hexafluorides such as tungsten hexafluoride (WF6). MoF6 is a colorless gas at room temperature and pressure, and it is highly reactive, particularly due to the strong electronegativity of fluorine atoms. The compound was first synthesized in 1947 by the reaction of molybdenum trioxide (MoO3) with elemental fluorine (F2). The synthesis of molybdenum hexafluoride involves a highly controlled reaction process. In the lab, molybdenum trioxide is reacted with elemental fluorine under specific conditions, such as high temperatures, to produce MoF6. The reaction is as follows: MoO3 + 3 F2 → MoF6 + O2 Molybdenum hexafluoride has applications in various chemical processes due to its strong oxidizing properties and ability to act as a fluorine source. It is primarily used in the production of other molybdenum compounds and in chemical vapor deposition (CVD) techniques. MoF6 is also used as a fluorinating agent in organic synthesis, where it can introduce fluorine atoms into organic molecules, providing valuable functionality in the development of pharmaceuticals and agrochemicals. One of the most significant uses of MoF6 is in the production of molybdenum-based alloys and materials for high-tech applications. Molybdenum is an essential metal in various industrial processes due to its strength and resistance to corrosion. The ability to introduce fluorine atoms into molybdenum compounds can enhance the properties of these materials, making them suitable for use in industries such as electronics, aerospace, and energy. Despite its reactivity and the risks associated with handling highly toxic and corrosive fluorine compounds, molybdenum hexafluoride remains a valuable reagent in the field of materials science. Researchers continue to explore its potential for the synthesis of advanced materials, and its role in the development of new fluorinated compounds remains an area of active investigation. However, due to its hazardous nature, safety protocols are critical when working with MoF6, particularly in industrial settings. In conclusion, molybdenum hexafluoride is an important chemical compound with a range of applications, from materials science to organic synthesis. Its discovery and subsequent use have enabled advancements in several fields, contributing to the development of high-performance materials and fluorinated compounds. Its continued exploration holds promise for further applications, although handling it requires caution due to its reactivity and toxicity. References 1962. A study of the fluorides of some multivalent metals by potentiometric titration in nonaqueous media. The Soviet Journal of Atomic Energy, 12(5). DOI: 10.1007/bf01591389 2016. Hydrogen reduction of 98MoF6 in RF discharge. Journal of Radioanalytical and Nuclear Chemistry, 309(2). DOI: 10.1007/s10967-015-4687-z 2020. Hydrogen Reduction of MoF6 and Molybdenum Carbide Formation in RF Inductively Coupled Low-Pressure Discharge: Experiment and Equilibrium Thermodynamics Consideration. Plasma Chemistry and Plasma Processing, 41(1). DOI: 10.1007/s11090-020-10138-3 |
| Market Analysis Reports |