Sodium formate, with the chemical formula HCOONa, is the sodium salt of formic acid. It is a white, odorless, and hygroscopic crystalline powder that is highly soluble in water and can also dissolve in alcohol. Sodium formate is utilized in various applications across different industries, including textiles, food, pharmaceuticals, and chemical manufacturing.
The discovery of sodium formate can be traced back to the early studies of formic acid, which was first isolated in 1670 by the British chemist John Ray from the stings of ants, as formic acid derives its name from the Latin word "formica," meaning ant. Sodium formate was later synthesized as researchers began to explore the chemical properties and reactions of formic acid and its salts. The formation of sodium formate typically occurs through the neutralization of formic acid with sodium hydroxide or sodium bicarbonate, leading to the production of sodium formate and water.
Sodium formate is widely used in the textile industry as a buffering agent and pH regulator in dyeing processes. Its ability to maintain a stable pH is crucial in ensuring uniform dye uptake in fabrics, thereby enhancing the quality and colorfastness of dyed textiles. Sodium formate also acts as a reducing agent in the dyeing process, promoting the formation of specific dye intermediates that yield desired colors.
In the food industry, sodium formate serves as a food preservative and a flavoring agent. It is recognized for its role in preventing the growth of certain bacteria and fungi, making it useful in extending the shelf life of various food products. Sodium formate is often used in meat processing, where it helps to improve the texture and flavor of cured meats. Additionally, it is used in the preparation of various food additives, contributing to the overall safety and quality of food products.
Sodium formate is also significant in the pharmaceutical industry, where it is employed as an excipient in the formulation of various medications. Its properties as a buffering agent and stabilizer are vital in maintaining the pH and overall stability of pharmaceutical preparations. Furthermore, sodium formate is utilized in the production of certain pharmaceutical intermediates, where it acts as a building block in the synthesis of complex organic molecules.
Another important application of sodium formate lies in chemical manufacturing. It is used as a reducing agent in various chemical reactions, including the synthesis of formic acid and other organic compounds. Sodium formate can also participate in decarboxylation reactions, leading to the formation of useful chemicals in organic synthesis. Moreover, it is utilized in the production of antifreeze and de-icing agents due to its low freezing point and ability to lower the freezing point of water.
In recent years, sodium formate has gained attention in environmental applications. It is being explored as a potential biodegradable alternative to conventional chemical agents in various processes, including wastewater treatment. Sodium formate can serve as a carbon source for microbial processes, promoting the degradation of organic pollutants and contributing to the overall treatment efficiency of wastewater.
While sodium formate is generally regarded as safe for use in food and pharmaceutical applications, it is essential to handle it with care. Exposure to large quantities can lead to irritation of the skin and eyes, and ingestion in significant amounts may result in gastrointestinal distress. Therefore, proper safety precautions, such as wearing gloves and protective eyewear, should be taken when handling sodium formate in industrial and laboratory settings.
In summary, sodium formate is a versatile chemical compound with a wide range of applications across various industries, including textiles, food, pharmaceuticals, and chemical manufacturing. Its discovery and subsequent utilization highlight the importance of formate salts in enhancing product quality and safety. The ongoing exploration of sodium formate's properties continues to unveil its potential in new and innovative applications.
References
1979. Complete degradation of carbohydrate to carbon dioxide and methane by syntrophic cultures of Acetobacterium woodii and Methanosarcina barkeri. Archives of Microbiology, 121(3).
DOI: 10.1007/bf00409211
2000. Intake, digestibility, and composition of orchardgrass and alfalfa silages treated with cellulase, inoculant, and formic acid fed to lambs. Journal of Animal Science, 78(11).
DOI: 10.2527/2000.78112980x
2024. Supercritical carbon dioxide likely served as a prebiotic source of methanethiol in primordial ocean hydrothermal systems. Communications Earth & Environment, 5(1).
DOI: 10.1038/s43247-024-01689-w
|






























































































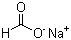
 GHS07 Warning Details
GHS07 Warning Details