Online Database of Chemicals from Around the World
| Shanghai Nanxiang Reagent Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nx-reagent.com | |||
 | +86 (21) 3912-7448 +86 13512139665 | |||
 | +86 (21) 3912-7431 | |||
 | xumiao@nx-reagent.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Nantong Chem-land Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chem-land.com | |||
 | +86 (513) 8551-2619 | |||
 | +86 (513) 8551-2676 | |||
 | sales@chem-land.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Changzhou Hangyu Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.czyys.com | |||
 | +86 (519) 8880-2789 +86 13961196887 | |||
 | +86 (519) 8880-2789 | |||
 | sales@czyys.com zypharm78@hotmail.com | |||
| Chemical manufacturer since 1984 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Silworld Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.silworldchemical.com | |||
 | +86 (27) 8561-3400 +86 15827173649 | |||
 | +86 (27) 8561-3419 | |||
 | info@silworldchemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Changzhou Litong Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.litonchem.com | |||
 | +86 (519) 8630-1238 | |||
 | +86 (519) 8659-1192 | |||
 | htszh2004@163.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Cloudscape Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cloudscapechem.com | |||
 | +86 (21) 6630-2580 | |||
 | +86 (21) 6631-4011 | |||
 | mollyqin@cschemsh.com molly0919@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shenzhen Vtolo Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.smt-tl.com | |||
 | +86 (755) 2767-0086 | |||
 | +86 (755) 2767-0086 | |||
 | antonia@vtolo.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Suqian Nanxiang Chemicals Manufacturing Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nanxiangchem.com | |||
 | +86 (0527) 8483-6748 | |||
 | sales@nanxiangchem.com | |||
 | QQ Chat | |||
 | WeChat: 13512139665 | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Auben Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aubenchem.com | |||
 | +86 19121901603 | |||
 | sales@aubenchem.com | |||
| Chemical distributor since 2023 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Acyclic carboxylic acid |
|---|---|
| Name | Malonic acid |
| Synonyms | 1,3-Propanedioic acid |
| Molecular Structure | 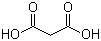 |
| Molecular Formula | C3H4O4 |
| Molecular Weight | 104.06 |
| CAS Registry Number | 141-82-2 |
| EC Number | 205-503-0 |
| SMILES | C(C(=O)O)C(=O)O |
| Density | 1.5±0.1 g/cm3, Calc.*, 1.631 g/mL (15 °C) |
|---|---|
| Melting Point | 134 °C |
| Index of Refraction | 1.479, Calc.* |
| Boiling Point | 386.8±25.0 °C (760 mmHg), Calc.*, 140 °C (760 mmHg) |
| Flash Point | 201.9±19.7 °C, Calc.*, 157 °C |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H318-H319 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Malonic acid, a dicarboxylic acid with the chemical formula C3H4O4, has played a significant role in organic chemistry since its discovery in the 19th century. It was first identified in the early 1800s by the French chemist Pierre Robiquet, who isolated it from the bitter almond oil, where it occurs naturally as part of various plant metabolic processes. The name "malonic" is derived from the Latin word "malon," meaning "apple," reflecting its presence in fruit. The structure of malonic acid features two carboxylic acid groups (-COOH) attached to a three-carbon backbone, making it a simple yet versatile compound. Its discovery paved the way for its use in various synthetic applications, particularly in the production of other organic compounds. One of the primary applications of malonic acid is as a key intermediate in organic synthesis. It is widely used in the malonic ester synthesis, where it reacts with various alkyl halides to form malonic esters. These esters serve as important building blocks in the preparation of α-substituted carboxylic acids, which are significant in pharmaceuticals and agrochemicals. The ability to introduce functional groups at the α-position of carboxylic acids through malonic acid derivatives makes it a valuable reagent in synthetic organic chemistry. Malonic acid also finds applications in the manufacture of polymers and resins. It can be polymerized to produce polyesters and polyamides, contributing to the production of various plastic materials with diverse applications, including coatings, adhesives, and fibers. Its role as a precursor in the synthesis of biodegradable polymers has gained attention in recent years, as environmental concerns drive the search for sustainable materials. Additionally, malonic acid has been explored for its potential health benefits and applications in the pharmaceutical industry. It is a precursor for the synthesis of certain pharmaceuticals, including some anti-inflammatory and analgesic agents. Its ability to participate in biochemical reactions has led researchers to investigate its role in cellular metabolism and energy production. In the field of agriculture, malonic acid has been utilized as a herbicide and fungicide. Its derivatives are being studied for their efficacy in controlling plant diseases and pests, providing an alternative to conventional agrochemicals. While malonic acid is generally regarded as safe, it is important to handle it with care, as exposure can lead to irritation of the skin and mucous membranes. Appropriate safety precautions should be taken when working with this compound in laboratory and industrial settings. In summary, malonic acid is a vital compound in organic chemistry, known for its versatile applications in synthesis, polymer production, and pharmaceuticals. Its historical significance and continued relevance underscore its importance in both academic research and industrial processes, making it a key player in the development of new materials and pharmaceuticals. References 2003. Cantharidin analogues: synthesis and evaluation of growth inhibition in a panel of selected tumour cell lines. Bioorganic Chemistry, 31(1). DOI: 10.1016/s0045-2068(02)00524-2 2003. Palladium-catalyzed alpha-arylation of carbonyl compounds and nitriles. Accounts of Chemical Research, 36(4). DOI: 10.1021/ar0201106 1984. Fluorometric determination of malonic acid with dicyclohexylcarbodiimide. Analytical Biochemistry, 140(1). DOI: 10.1016/0003-2697(84)90153-2 |
| Market Analysis Reports |