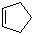
Cyclopentene is an unsaturated cyclic hydrocarbon with the molecular formula C5H8. It consists of a five-membered carbon ring containing one double bond, making it a monocyclic alkene. Cyclopentene is a colorless liquid at room temperature with a mild, slightly sweet odor. It is flammable and volatile, and it is soluble in organic solvents such as ether, alcohol, and benzene but only sparingly soluble in water.
The compound was first identified and isolated in the early developments of hydrocarbon chemistry in the 19th century, with systematic study following improvements in distillation and analytical techniques. Cyclopentene can be produced through various synthetic routes, including the dehydration of cyclopentanol using acid catalysts, or by the cracking of petroleum fractions under specific conditions. Industrially, it may also be formed during pyrolysis processes or recovered from the complex mixtures of hydrocarbons in naphtha.
Cyclopentene is an important intermediate in organic synthesis and industrial chemistry. One of its most prominent applications is as a starting material for the synthesis of cyclopentane, which is used as a blowing agent in the manufacture of polyurethane foams. Hydrogenation of cyclopentene over metal catalysts such as palladium or platinum leads to the formation of cyclopentane under mild conditions.
In polymer chemistry, cyclopentene serves as a monomer for ring-opening metathesis polymerization (ROMP). This reaction is catalyzed by transition metal complexes such as those of molybdenum or ruthenium, resulting in polypentenamers—unsaturated polymers that can be chemically modified or hydrogenated for diverse material applications. These polymers are valued for their flexibility, thermal stability, and potential use in specialty elastomers and coatings.
Cyclopentene is also used as a synthetic building block in the preparation of various fine chemicals, pharmaceuticals, and agrochemicals. Its reactivity arises from the presence of the double bond, which can participate in addition reactions, including halogenation, hydrohalogenation, epoxidation, and dihydroxylation. Functionalized derivatives of cyclopentene are common intermediates in complex organic syntheses, including the synthesis of natural products and heterocycles.
In research, cyclopentene has been studied as a substrate in Diels–Alder reactions and other pericyclic processes. Its cyclic structure and unsaturation make it suitable for exploring reaction mechanisms involving strain, regioselectivity, and stereoselectivity. It also plays a role in studying photochemical and radical reactions involving alkenes.
From an environmental and safety standpoint, cyclopentene is classified as a hazardous substance. It is highly flammable, and its vapors can form explosive mixtures with air. Exposure to high concentrations may cause respiratory irritation, dizziness, or drowsiness. Proper storage in tightly sealed containers away from sources of ignition, heat, and oxidizing agents is essential.
In summary, cyclopentene is a volatile, flammable five-membered cyclic alkene with broad applications in industrial and synthetic chemistry. It is used as a precursor to cyclopentane, a monomer for polymer synthesis, and a versatile intermediate in the preparation of fine chemicals. Its chemical reactivity, particularly its susceptibility to electrophilic addition and polymerization, makes it a valuable compound in both applied and academic chemistry.
References
2024. The π-π stacking effect of metallocene catalyst in olefin polymerization. Polymer Bulletin, 81(14).
DOI: 10.1007/s00289-024-05432-5
2024. Recent advances in metal-mediated oxidations with mCPBA. Transition Metal Chemistry, 49(6).
DOI: 10.1007/s11243-024-00593-8
1994. Synthesis of steroidal D-ring fused pyrazolines: Study of regiochemistry of addition. Steroids, 59(8).
DOI: 10.1016/0039-128x(94)90061-2
|





















































 GHS02;GHS07;GHS08 Danger Details
GHS02;GHS07;GHS08 Danger Details