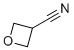
Oxetane-3-carbonitrile is a small, strained heterocyclic compound featuring a four-membered oxetane ring substituted at the 3-position with a nitrile (-CN) group. The oxetane ring consists of three carbon atoms and one oxygen atom, forming a highly strained cyclic ether that is known for its unique chemical and physicochemical properties. The presence of the nitrile substituent introduces an electron-withdrawing group that significantly alters the electronic profile of the ring system, enhancing its reactivity and synthetic versatility.
The synthesis of oxetane-3-carbonitrile can be achieved through several established methods. One common approach involves nucleophilic substitution of a 3-halooxetane derivative with a cyanide source such as sodium cyanide or potassium cyanide. Another strategy includes the ring closure of a β-hydroxy nitrile under intramolecular cyclization conditions, using strong bases or dehydrating agents. These methods allow for efficient preparation of oxetane-3-carbonitrile in both laboratory and industrial settings.
The oxetane ring system is well known for its compact size, ring strain, and ability to modulate physicochemical properties such as polarity, lipophilicity, and metabolic stability. When incorporated into drug-like molecules, oxetanes often act as isosteric replacements for gem-dimethyl, carbonyl, or ether groups, helping to improve bioavailability and reduce off-target metabolism. The nitrile group, on the other hand, provides a site for further chemical transformations, including reductions to primary amines, hydrolysis to carboxylic acids or amides, and participation in multicomponent reactions such as the Ugi reaction.
Oxetane-3-carbonitrile has found increasing application in medicinal chemistry as a versatile fragment in fragment-based drug discovery (FBDD). Its rigid, three-dimensional structure and small size make it an attractive scaffold for optimizing binding interactions with biological targets. Additionally, the combination of ring strain and electron-withdrawing nitrile functionality makes the molecule reactive under mild conditions, enabling rapid derivatization and structure–activity relationship (SAR) development.
In the broader context of materials and synthetic chemistry, oxetane derivatives are also studied for their potential use in photopolymerization and the formation of crosslinked polymers, although oxetane-3-carbonitrile itself is more typically associated with small-molecule applications.
In summary, oxetane-3-carbonitrile is a synthetically accessible, chemically reactive compound of growing interest in medicinal and synthetic chemistry. Its unique structural features, including the strained oxetane ring and polar nitrile group, provide valuable opportunities for structural modification and functional diversification in drug development and beyond.
|




 GHS06 Danger Details
GHS06 Danger Details