Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2010 | ||||
| Classification | Organic raw materials >> Heterocyclic compound |
|---|---|
| Name | (3R,4S)-3-(2-Bromoacetyl)-4-ethyl-1-pyrrolidinecarboxylic acid phenylmethyl ester |
| Synonyms | benzyl (3R,4S)-3-(2-bromoacetyl)-4-ethylpyrrolidine-1-carboxylate |
| Molecular Structure | 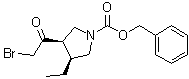 |
| Molecular Formula | C16H20BrNO3 |
| Molecular Weight | 354.24 |
| CAS Registry Number | 1428243-26-8 |
| SMILES | CC[C@@H]1CN(C[C@@H]1C(=O)CBr)C(=O)OCC2=CC=CC=C2 |
| Solubility | Practically insoluble (0.043 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.361±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2017 ACD/Labs) |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details |
|---|---|
| Risk Statements | H302-H314 Details |
| Safety Statements | P280-P305+P351+P338-P310 Details |
| SDS | Available |
|
(3R,4S)-3-(2-bromoacetyl)-4-ethyl-1-pyrrolidinecarboxylic acid benzyl ester is an interesting compound that has attracted much attention in organic and medicinal chemistry. The synthesis of this compound focuses on creating chiral molecules with specific functional groups. Its structure includes a pyrrolidine ring with a 2-bromoacetyl group, an ethyl substituent, and a benzyl ester. This arrangement not only makes the compound structurally unique, but also enhances its reactivity and potential interactions with biological targets. In medicinal chemistry, the therapeutic potential of (3R,4S)-3-(2-bromoacetyl)-4-ethyl-1-pyrrolidinecarboxylic acid benzyl ester is being explored. The reactivity introduced by the bromoacetyl group can be used in drug development, especially for designing molecules that interact with specific biological pathways. Its unique structure makes it a candidate for investigation of new treatments for various diseases, including those that require precise molecular interactions. In addition to its medicinal applications, this compound has several other potential uses. It is an important intermediate in organic synthesis and helps in the creation of more complex molecules. Its stereochemistry can be used in asymmetric synthesis to produce enantiomerically pure compounds. The effects of this compound on neurotransmitter systems have been studied, which could help in the study of neurological diseases. In materials science, the chemical properties of this compound can be used to develop new materials such as specialized polymers or coatings. Its reactive groups make it useful for labeling or attaching molecules for imaging or drug delivery. References 2024. Sensitive and selective determination of upadacitinib using a custom-designed electrochemical sensor based on ZnO nanoparticle-assisted molecularly imprinted polymer. Analytical and Bioanalytical Chemistry, 416, 22. DOI: 10.1007/s00216-024-05541-0 2022. Synthesis of Upadacitinib. Synfacts, 18, 6. DOI: 10.1055/s-0041-1738394 2020. Upadacitinib. Pharmaceutical Substances, 1, 1. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-21-0010 |
| Market Analysis Reports |