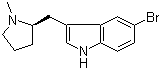
(R)-5-Bromo-3-((1-methylpyrrolidin-2-yl)methyl)-1H-indole is a chiral indole derivative that falls within a class of compounds known for their pharmacological relevance, particularly in neurochemistry and medicinal chemistry. Its structure combines an indole core—a scaffold found in many biologically active natural products and drugs—with a chiral 1-methylpyrrolidin-2-ylmethyl substituent at the 3-position and a bromine atom at the 5-position. The (R)-configuration at the pyrrolidine ring carbon bearing the methyl group contributes to its stereoselective biological activity, a key consideration in drug design.
Indole derivatives, especially those substituted at the 3-position, have a broad range of biological activities. The substitution of the pyrrolidine moiety introduces a nitrogen-containing heterocycle known to affect receptor binding and metabolic stability. The presence of the 5-bromo substituent on the aromatic ring can influence the compound’s electronic properties, receptor affinity, and overall pharmacokinetics. Halogen substitution in aromatic systems is often used to modulate lipophilicity and facilitate interactions with binding pockets in protein targets.
The development of compounds such as (R)-5-bromo-3-((1-methylpyrrolidin-2-yl)methyl)-1H-indole is often linked to studies of neurotransmitter systems, especially the serotonin (5-HT) receptor subtypes, due to the indole’s structural similarity to serotonin itself. Derivatives of this type have been investigated for their binding affinities to 5-HT2A, 5-HT1A, and other central nervous system receptors. The stereochemistry plays a pivotal role in binding interactions, as enantiomers may exhibit different potency, efficacy, or receptor subtype selectivity.
Research in the area of synthetic tryptamines, which are structurally related to this compound, has explored various analogs as potential tools for neuroscience research, as well as for therapeutic applications in psychiatry. The design of chiral indole derivatives also aligns with trends in precision pharmacology, where receptor-specific and stereoselective ligands are desirable for minimizing side effects and improving clinical outcomes.
Synthetic routes to such compounds typically begin with functionalized indole precursors, where a halogen (such as bromine) is introduced through electrophilic aromatic substitution or halogen exchange reactions. The chiral pyrrolidine moiety may be incorporated through alkylation strategies involving enantiomerically enriched intermediates or by using chiral auxiliaries or catalysts to control the configuration at the stereocenter.
Applications of compounds with this scaffold extend to the study of G protein-coupled receptors (GPCRs), monoamine oxidase inhibition, and as ligands in molecular imaging. Depending on further substitution and structural modification, such compounds can act as agonists, antagonists, or modulators, contributing to both therapeutic and diagnostic developments.
(R)-5-Bromo-3-((1-methylpyrrolidin-2-yl)methyl)-1H-indole is one example among many in the broader category of indole-based molecules engineered to target central nervous system pathways. Its unique structure offers opportunities for selective interaction with biological targets, making it valuable for research into neuroactive drugs and receptor pharmacology.
References
none
|












































 GHS05;GHS07;GHS08;GHS09 Danger Details
GHS05;GHS07;GHS08;GHS09 Danger Details