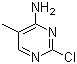
4-Amino-2-chloro-5-methylpyrimidine is a heteroaromatic compound belonging to the pyrimidine family, which has been of central importance in both natural and synthetic chemistry. The discovery of this substance was linked to broader investigations into substituted pyrimidines in the early and mid-20th century, a period when chemists were systematically studying the modification of pyrimidine scaffolds to explore their chemical properties and biological activities. Substituents at the 2-, 4-, and 5-positions of the pyrimidine ring were of particular interest because of their relevance to nucleobase analogues and their potential to serve as intermediates in the preparation of more complex compounds. The introduction of chlorine at the 2-position combined with an amino group at the 4-position provided a versatile platform for chemical derivatization, while the methyl group at the 5-position contributed additional steric and electronic effects. This made 4-amino-2-chloro-5-methylpyrimidine a compound of practical synthetic value.
Applications of 4-amino-2-chloro-5-methylpyrimidine have been largely centered on its role as an intermediate in pharmaceutical and agrochemical synthesis. The 2-chloro substituent functions as a reactive leaving group, making the compound suitable for nucleophilic substitution reactions. Through displacement of chlorine by various nucleophiles, chemists have been able to generate a wide range of substituted pyrimidines, many of which exhibit biological activity. This property has been particularly useful in medicinal chemistry, where the pyrimidine core is a privileged scaffold in the design of antiviral, anticancer, and antibacterial agents. The amino group at the 4-position provides further opportunities for hydrogen bonding and derivatization, increasing its value as a starting material.
In pharmaceutical research, derivatives of 4-amino-2-chloro-5-methylpyrimidine have been studied for their potential as enzyme inhibitors and receptor ligands. Substitution reactions on this compound can yield analogues of nucleobases that interfere with DNA or RNA synthesis, a strategy employed in the development of certain classes of antiviral and anticancer drugs. Although the compound itself is not used therapeutically, it has enabled the synthesis of more complex molecules with pharmacological significance. Its reactivity has also been exploited in the creation of kinase inhibitors and compounds targeting metabolic enzymes, areas where pyrimidine chemistry continues to play an important role.
In agrochemical research, the compound has been applied in the synthesis of herbicides and fungicides. Substituted pyrimidines derived from it have been shown to affect biochemical pathways in plants and fungi, making them useful as crop protection agents. The combination of an amino group and a methyl substituent on the pyrimidine ring often enhances biological interaction by modifying lipophilicity and electronic distribution. As a result, 4-amino-2-chloro-5-methylpyrimidine has served as a reliable precursor in the preparation of compounds that contribute to agricultural productivity.
The discovery and continued use of 4-amino-2-chloro-5-methylpyrimidine underscore the enduring significance of pyrimidine chemistry. Its structural features provide both reactivity and versatility, making it a practical tool in synthetic chemistry. Through its applications in the development of pharmaceuticals and agrochemicals, the compound has contributed to advances in health and agriculture, reflecting the central role of heteroaromatic intermediates in modern applied chemistry.
References
2020. Tailor-made amino acid-derived pharmaceuticals approved by the FDA in 2019. Amino Acids, 52(9).
DOI: 10.1007/s00726-020-02887-4
1970. 1,3-Dimercaptopropyl-2-thiopyrimidines. Chemistry of Heterocyclic Compounds, 6(5).
DOI: 10.1007/bf00500672
1968. Über Pyrimidinderivate. Monatshefte für Chemie - Chemical Monthly, 99(5).
DOI: 10.1007/bf00901240
|



































 GHS05;GHS06;GHS07 Danger Details
GHS05;GHS06;GHS07 Danger Details