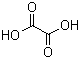
Oxalic acid is a naturally occurring dicarboxylic acid with the molecular formula C2H2O4. It exists in both anhydrous and dihydrate forms, with the latter being more common and widely used. Structurally, it contains two carboxyl groups directly bonded to each other, making it the simplest of all dicarboxylic acids. The compound is a white crystalline solid, soluble in water, and exhibits strong acidity, making it a versatile reagent in chemical and industrial applications.
The compound was first isolated in the 18th century through the oxidation of organic matter, and in 1776, Swedish chemist Carl Wilhelm Scheele succeeded in isolating oxalic acid from sorrel and rhubarb. It was later synthesized from sugar using nitric acid oxidation, and this laboratory synthesis marked one of the earliest examples of converting plant-derived material into a defined organic compound. This milestone played an important role in the historical development of organic chemistry and the understanding of carboxylic acid chemistry.
Oxalic acid is widely distributed in nature. It is found in many plants, including spinach, sorrel, beet greens, rhubarb, and tea leaves. In plants, it is typically present as a salt called oxalate, often complexed with calcium. In biological systems, oxalates are formed as metabolic byproducts and can contribute to the formation of kidney stones in the form of calcium oxalate under certain pathological conditions. Due to its chelating ability, oxalic acid can bind metal ions, a property that underlies many of its applications.
One of the major industrial uses of oxalic acid is as a cleaning and bleaching agent. Its ability to chelate iron, calcium, and other metal ions makes it effective for removing rust and scale from metals, as well as for cleaning minerals and stone surfaces. In textile processing, it is used for bleaching and dye stripping. In the leather industry, oxalic acid is applied in the cleaning and finishing of hides. Additionally, it serves as a mordant in dyeing processes, where it helps fix dyes to fibers by forming coordination complexes with dye molecules and fiber-bound metals.
In analytical chemistry, oxalic acid is used as a primary standard for the calibration of solutions in redox titrations, particularly those involving potassium permanganate. Its reliable and reproducible redox behavior makes it suitable for quantitative chemical analysis. It is also employed in metal polishing formulations, wood cleaning products, and as an intermediate in the synthesis of pharmaceuticals, rare earth separation agents, and other fine chemicals.
Oxalic acid has been studied as a reagent in organic synthesis, especially in reactions requiring mild reducing conditions. It can decompose upon heating or under acidic conditions to produce carbon monoxide, carbon dioxide, and formic acid. These properties are harnessed in specialized laboratory procedures and research settings. Moreover, due to its ability to form stable metal complexes, oxalic acid has found use in metallurgical processes for selective leaching and precipitation of metals.
Safety considerations are important in the use of oxalic acid. While it occurs naturally in food plants, the pure acid is toxic if ingested and can cause severe irritation or damage upon contact with skin and mucous membranes. Proper handling, storage, and disposal are essential to minimize risk. Chronic exposure to oxalic acid or its salts may lead to calcium depletion in the body and the formation of kidney stones.
Despite its toxicity, oxalic acid continues to play an essential role in industrial, laboratory, and research applications. Its discovery and utility illustrate the chemical importance of simple organic acids and their multifaceted roles across scientific and technological domains.
References
Riemenschneider W, Tanifuji M (2000) Oxalic Acid Ullmann's Encyclopedia of Industrial Chemistry Wiley-VCH DOI: 10.1002/14356007.a18_247
Dean P A W (2012) The Oxalate Dianion, C2O42-: Planar or Nonplanar? Journal of Chemical Education 89(3) 417–418 DOI: 10.1021/ed200202r
Gupta VK et al. (2012) Chemical treatment technologies for waste‑water recycling—an overview RSC Advances 2(16) 6380–6388 DOI: 10.1039/C2RA20340E
|












































































 GHS07 Warning Details
GHS07 Warning Details