Online Database of Chemicals from Around the World
| Zhohoua Biotech PVT | India | |||
|---|---|---|---|---|
 | zhbiotech.com | |||
 | +91 9319668540 | |||
 | +91 9319668540 | |||
 | zhohouabiotech@zohomail.com | |||
 | Skype Chat | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | API >> Nervous system medication >> Other nervous system medication |
|---|---|
| Name | Cyclazodone |
| Synonyms | 2-(Cyclopropylamino)-5-Phenyl-Oxazol-4-One; 2-(Cyclopropylamino)-5-Phenyl-4-Oxazolone; Cyclexanone |
| Molecular Structure | 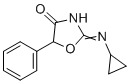 |
| Molecular Formula | C12H12N2O2 |
| Molecular Weight | 216.24 |
| CAS Registry Number | 14461-91-7 |
| EC Number | 111-755-1 |
| SMILES | C1CC1N=C2NC(=O)C(O2)C3=CC=CC=C3 |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 334.8±45.0 °C 760 mmHg (Calc.)* |
| Flash point | 156.3±28.7 °C (Calc.)* |
| Index of refraction | 1.688 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS06 Danger Details GHS06 Danger Details |
|---|---|
| Risk Statements | H301 Details |
| Safety Statements | P264-P270-P301+P316-P321-P330-P405-P501 Details |
|
Cyclazodone (systematic name: 2-(cyclopropylmethyl)-4,4-dimethyl-3-(2-oxo-1-pyrrolidinyl)-2-oxazolidinone) is a synthetic compound belonging to the oxazolidinone class. It was first reported in the 1960s during investigations into central nervous system stimulants structurally related to pemoline. Cyclazodone was developed as part of efforts to create compounds with psychostimulant and potential nootropic properties while attempting to reduce some of the side effects associated with earlier agents in the same family. The compound was synthesized through structural modifications of pemoline, replacing the oxazolidinone moiety with various substituents to evaluate pharmacological activity. Cyclazodone was found to act as a central nervous system stimulant in animal studies, showing activity in tests commonly used to screen for psychostimulant effects, such as increased locomotor activity. Compared to pemoline, it was observed to possess greater potency in certain assays, although detailed pharmacological characterization in humans has been limited. Cyclazodone has not been widely adopted for clinical use and remains a research chemical. Unlike pemoline, which was once marketed as a treatment for attention-deficit hyperactivity disorder (ADHD) and narcolepsy before being withdrawn due to concerns about hepatotoxicity, cyclazodone was never developed into a commercial pharmaceutical. Its pharmacokinetics, metabolism, and toxicity profile in humans have not been comprehensively studied, and therefore it is not approved for medical use by regulatory authorities. The applications of cyclazodone have been limited to research contexts, primarily within medicinal chemistry investigations of oxazolidinone derivatives and their effects on the central nervous system. Its reported stimulant properties have made it of interest in studies aiming to better understand structure–activity relationships among heterocyclic compounds with psychostimulant potential. However, the lack of thorough clinical evaluation and safety data has prevented its development into an approved therapeutic agent. Cyclazodone remains a niche compound in the history of stimulant research. While it exemplifies the chemical strategies used in the mid-20th century to design and evaluate new psychoactive agents, it did not advance beyond preliminary pharmacological testing. Today, it is primarily cited in chemical and pharmacological literature as an example of oxazolidinone-based stimulants related to pemoline. References 2010. Multigram Synthesis of trans-2-(Trifluoromethyl)cyclopropanamine. Synthesis, 2010(24). DOI: 10.1055/s-0030-1258323 1966. Comptes Rendus des Seances de l'Academie des Sciences, Serie D: Sciences Naturelles, 262(213). PMID: 4379379 British Patent Document, #1005738. |
| Market Analysis Reports |