Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Changzhou Lanming Science and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lmkjcz.com | |||
 | +86 (519) 8881-6918 +86 13906110072 +86 13915000734 | |||
 | +86 (519) 8825-5676 | |||
 | lmkj@lmkjcz.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Great Forest Biomedical Ltd. | China | |||
|---|---|---|---|---|
 | www.great-forest.com | |||
 | +86 (571) 2831-3180 | |||
 | +86 (571) 2831-3182 | |||
 | marketing@great-forest.com info@great-forest.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Changzhou Odoc International Trade Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.odoc-co.com | |||
 | +86 (519) 8515-5812 | |||
 | +86 (519) 8515-5813 | |||
 | office1@odoc-co.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Coresyn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.coresyn.com | |||
 | +86 (571) 8662-6709 | |||
 | sales@coresyn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai CR Corporation Limited | China | |||
|---|---|---|---|---|
 | www.crcorporation.cn | |||
 | +86 (21) 5736-6208 +86 13917420128 | |||
 | +86 (21) 5736-6228 | |||
 | fred.wen@crcorporation.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Aaron Chemistry GmbH | Germany | |||
|---|---|---|---|---|
 | www.aaron-chemistry.de | |||
 | +49 (8823) 917-521 | |||
 | +49 (8823) 917-523 | |||
 | sales@aaron-chemistry.de | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Spectrum Info Ltd. | Ukraine | |||
|---|---|---|---|---|
 | www.spec-info.com | |||
 | +38 (044) 573-5462 | |||
 | +38 (044) 573-2397 | |||
 | spec-info@spec-info.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> API intermediate |
|---|---|
| Name | 3-Methylpyrazole |
| Synonyms | 5-methyl-1H-pyrazole |
| Molecular Structure | 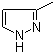 |
| Molecular Formula | C4H6N2 |
| Molecular Weight | 82.10 |
| CAS Registry Number | 1453-58-3 |
| EC Number | 215-925-7 |
| SMILES | CC1=CC=NN1 |
| Density | 1.1±0.1 g/cm3 Calc.*, 1.017 g/mL (Expl.) |
|---|---|
| Melting point | 36.5 °C (Expl.) |
| Boiling point | 204.0 °C 760 mmHg (Calc.)*, 203 - 205 °C (Expl.) |
| Flash point | 87.4±11.7 °C (Calc.)*, 93 °C (Expl.) |
| Index of refraction | 1.523 (Calc.)*, 1.496 (Expl.) |
| Water solubility | miscible |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS05;GHS07;GHS08 Danger Details GHS05;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H314:-H318:-H360D:-H373: Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P264-P264+P265-P270-P280-P301+P317-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P318-P319-P321-P330-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
3-Methylpyrazole is a heterocyclic organic compound with the molecular formula C4H6N2. It is a substituted pyrazole, consisting of a five-membered aromatic ring containing two adjacent nitrogen atoms (at positions 1 and 2) and a methyl group at the 3-position. This structure imparts both basic and nucleophilic properties to the molecule, which appears as a colorless to pale yellow liquid under ambient conditions and has a characteristic amine-like odor. The discovery of pyrazole derivatives dates back to the 19th century during systematic investigations of nitrogen-containing heterocycles. 3-Methylpyrazole itself can be synthesized by the condensation of hydrazine with 3-methyl-1,3-dicarbonyl compounds or β-ketoesters under acidic or basic conditions. A common laboratory route involves cyclization of 3-methyl-1-phenyl-1,3-propanedione with hydrazine hydrate, yielding the desired pyrazole derivative after work-up and purification. 3-Methylpyrazole has gained particular attention for its biological and pharmaceutical relevance. It is best known for its use as a potent inhibitor of alcohol dehydrogenase (ADH), the enzyme responsible for oxidizing ethanol to acetaldehyde in the human liver. This property is the basis for its use in medicine, particularly in the treatment of methanol or ethylene glycol poisoning. When administered, 3-methylpyrazole (under the generic name fomepizole) competitively inhibits ADH, slowing the formation of toxic metabolites and allowing time for elimination of the parent compounds from the body. In this medical application, 3-methylpyrazole is administered intravenously and is generally well tolerated when used in recommended doses. It offers a safer alternative to ethanol therapy, which was traditionally used to compete with methanol or ethylene glycol for ADH binding. Unlike ethanol, 3-methylpyrazole has fewer central nervous system effects and a more predictable pharmacokinetic profile, making it a preferred antidote in clinical toxicology. Outside of its therapeutic use, 3-methylpyrazole serves as a building block in organic synthesis. Its ring system is a common motif in medicinal chemistry and agrochemical development due to its aromaticity and capacity to engage in hydrogen bonding and coordination with metal centers. It is used as a ligand in coordination chemistry, particularly in the synthesis of metal complexes where the nitrogen atoms of the pyrazole ring act as donor sites. These complexes can exhibit useful catalytic, magnetic, or electronic properties. In industrial applications, 3-methylpyrazole is used in the production of specialty chemicals, intermediates for pharmaceuticals, and heterocyclic compounds used in research. It can be modified through electrophilic substitution reactions, N-alkylation, and other functionalization methods to introduce desired properties for chemical libraries or structure–activity relationship (SAR) studies. 3-Methylpyrazole is also utilized in analytical chemistry, including the formulation of derivatization agents and as a component in reagent kits. In laboratory settings, it can serve as a model compound for studying the behavior of pyrazole derivatives under various chemical and physical conditions. Standard safety precautions apply when handling 3-methylpyrazole. Although it has relatively low acute toxicity, it can cause irritation upon contact with the skin or mucous membranes. Proper storage includes keeping the substance in tightly closed containers in a well-ventilated, cool, and dry environment, away from strong oxidizers or acids. In summary, 3-methylpyrazole is a substituted aromatic heterocycle with important roles in both medical and chemical applications. It is most notably used as an antidote for alcohol poisoning through its inhibition of alcohol dehydrogenase and also serves as a versatile synthetic intermediate, ligand, and pharmacophore in various areas of research and industry. References 2024. Green Synthesis of Alkenes as Antimicrobial Agents from the Corresponding Formyl Pyrazoles in Aqueous Medium and Insights from Molecular Docking. Chemistry Africa, 7(8). DOI: 10.1007/s42250-024-01054-6 2024. Trifluoroacetaldehyde N-tosylhydrazone in [3+2] cycloaddition reaction for the synthesis of 5-(trifluoromethyl)pyrazolines. Russian Chemical Bulletin, 73(4). DOI: 10.1007/s11172-024-4215-9 2021. Quinolinyl-pyrazoles: synthesis and pharmacological evolution in the recent decennial. Journal of the Iranian Chemical Society, 18(12). DOI: 10.1007/s13738-020-02152-1 |
| Market Analysis Reports |