Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2010 | ||||
| Classification | API >> Urinary system medication >> Diabetes insipidus medication |
|---|---|
| Name | Terlipressin |
| Synonyms | N-(N-(N-Glycylglycyl)glycyl)-8-L-lysinevasopressin; (2S)-1-[(4S,7S,10S,13S,16S,19S)-19-[[2-[[2-[(2-Aminoacetyl)amino]acetyl]amino]acetyl]amino]-13-benzyl-10-(2-carbamoylethyl)-7-(carbamoylmethyl)-16-[(4-hydroxyphenyl)methyl]-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentazacycloicosane-4-carbonyl]-N-[(1S)-5-amino-1-(carbamoylmethylcarbamoyl)pentyl]pyrrolidine-2-carboxamide |
| Molecular Structure | 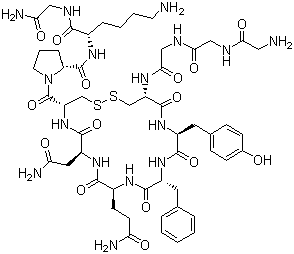 |
| Molecular Formula | C52H74N16O15S2 |
| Molecular Weight | 1227.37 |
| Protein Sequence | GGGCYFQNCPKG |
| CAS Registry Number | 14636-12-5 |
| EC Number | 238-680-8 |
| SMILES | C1C[C@H](N(C1)C(=O)[C@@H]2CSSC[C@@H](C(=O)N[C@H](C(=O)N[C@H](C(=O)N[C@H](C(=O)N[C@H](C(=O)N2)CC(=O)N)CCC(=O)N)CC3=CC=CC=C3)CC4=CC=C(C=C4)O)NC(=O)CNC(=O)CNC(=O)CN)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N |
| Density | 1.5±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 1824.0±65.0 °C 760 mmHg (Calc.)* |
| Flash point | 1056.9±34.3 °C (Calc.)* |
| Index of refraction | 1.664 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H332 Details | ||||||||
| Safety Statements | P261-P271-P304+P340-P317 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
Terlipressin is a synthetic analog of vasopressin designed to act primarily on V1 adrenergic receptors, inducing vasoconstriction in vascular smooth muscle. It is structurally modified from arginine vasopressin by the addition of three glycyl residues at the N-terminal, which prolongs its duration of action and reduces its immediate vasoconstrictive potency compared to native vasopressin. This modification allows terlipressin to provide sustained hemodynamic effects with less frequent dosing. The compound was developed to manage conditions associated with systemic and splanchnic vasodilation, such as hepatorenal syndrome, esophageal variceal bleeding, and septic shock. By stimulating V1 receptors on vascular smooth muscle, terlipressin increases systemic vascular resistance and mean arterial pressure, while its relative sparing of V2 receptor activity reduces water retention and minimizes the risk of hyponatremia. The controlled vasoconstrictive action makes terlipressin particularly effective in reducing portal hypertension and controlling acute variceal hemorrhage in cirrhotic patients. Terlipressin is typically administered intravenously or as an intravenous bolus, where it is rapidly converted to lysine vasopressin, the active form, through enzymatic cleavage. Its pharmacokinetics demonstrate a prolonged half-life relative to vasopressin, allowing sustained receptor stimulation and improved clinical management of hemodynamic instability. In the treatment of hepatorenal syndrome, terlipressin combined with albumin has been shown to improve renal perfusion and increase urine output, thereby reversing functional kidney failure associated with advanced liver disease. Clinical trials have demonstrated that terlipressin effectively reduces mortality and complications in patients with variceal bleeding. It is often used as a first-line pharmacological therapy to control bleeding while patients await endoscopic intervention. Furthermore, the peptide’s selective vasoconstrictive profile has been explored in septic shock and other hypotensive states, where it may help restore vascular tone without excessive fluid administration. Common adverse effects include abdominal cramps, ischemic events in peripheral tissues, and, less frequently, arrhythmias. Careful dosing and monitoring are recommended, particularly in patients with cardiovascular comorbidities. The development of terlipressin highlights the potential of peptide analogs of endogenous hormones for targeted therapeutic use. By mimicking the physiological actions of vasopressin while extending duration of action and improving safety, terlipressin exemplifies rational drug design in endocrine pharmacology. Its clinical utility in acute and chronic conditions demonstrates how synthetic peptides can be leveraged to achieve specific receptor-mediated effects, providing important therapeutic options in hepatology, critical care, and emergency medicine. References REVERSE Study Investigators (2016) Terlipressin plus albumin is more effective than albumin alone in improving renal function in patients with cirrhosis and hepatorenal syndrome type 1. Gastroenterology 150(7) 1579–1589.e2 DOI: 10.1053/j.gastro.2016.02.026 Wong FM, Pappas SC, Curry MP, Reddy KR, Rubin RA, Porayko MK, et al. (2021) Terlipressin plus albumin for the treatment of type 1 hepatorenal syndrome. New England Journal of Medicine 384(9) 818–828 DOI: 10.1056/NEJMoa2008290 Angeli P, Gines P (2017) Reversal of hepatorenal syndrome type 1 with terlipressin plus albumin: pooled analysis of phase 3 studies. Alimentary Pharmacology & Therapeutics 45(11) 1390–1402 DOI: 10.1111/apt.14052 |
| Market Analysis Reports |