Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2010 | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Other protected amino acids |
|---|---|
| Name | Teriparatide acetate |
| Synonyms | (4S)-4-[[(2S)-2-[[(2S)-2-[[(2S)-4-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-6-amino-2-[[2-[[(2S)-2-[[(2S)-4-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-5-amino-2-[[(2S,3S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-amino-3-hydroxypropanoyl]amino]-3-methylbutanoyl]amino]-3-hydroxypropanoyl]amino]-4-carboxybutanoyl]amino]-3-methylpentanoyl]amino]-5-oxopentanoyl]amino]-4-methylpentanoyl]amino]-4-methylsulfanylbutanoyl]amino]-3-(1H-imidazol-4-yl)propanoyl]amino]-4-oxobutanoyl]amino]-4-methylpentanoyl]amino]acetyl]amino]hexanoyl]amino]-3-(1H-imidazol-4-yl)propanoyl]amino]-4-methylpentanoyl]amino]-4-oxobutanoyl]amino]-3-hydroxypropanoyl]amino]-4-methylsulfanylbutanoyl]amino]-5-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-6-amino-1-[[(2S)-6-amino-1-[[(2S)-1-[[(2S)-5-amino-1-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-4-amino-1-[[(1S)-1-carboxy-2-phenylethyl]amino]-1,4-dioxobutan-2-yl]amino]-3-(1H-imidazol-4-yl)-1-oxopropan-2-yl]amino]-3-methyl-1-oxobutan-2-yl]amino]-3-carboxy-1-oxopropan-2-yl]amino]-1,5-dioxopentan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-1-oxohexan-2-yl]amino]-1-oxohexan-2-yl]amino]-5-carbamimidamido-1-oxopentan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-3-(1H-indol-3-yl)-1-oxopropan-2-yl]amino]-4-carboxy-1-oxobutan-2-yl]amino]-3-methyl-1-oxobutan-2-yl]amino]-5-carbamimidamido-1-oxopentan-2-yl]amino]-5-oxopentanoic acid |
| Molecular Structure | 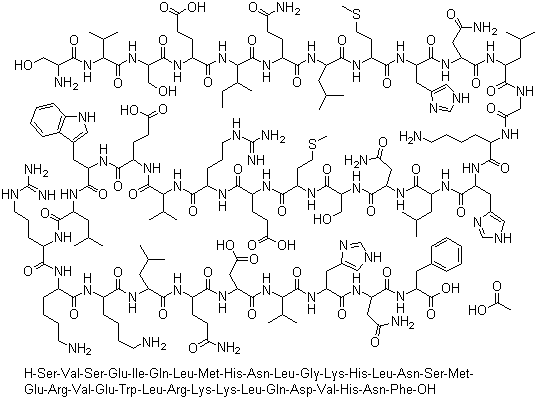 |
| Molecular Formula | C181H291N55O51S2.C2H4O2 |
| Molecular Weight | 4177.77 |
| Protein Sequence | SVSEIQLMHNLGKHLNSMERVEWLRKKLQDVHNF |
| CAS Registry Number | 52232-67-4 |
| EC Number | 640-978-1 |
| SMILES | CC[C@H](C)[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC2=CNC=N2)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CCCNC(=N)N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CC3=CNC4=CC=CC=C43)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(=N)N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC5=CNC=N5)C(=O)N[C@@H](CC(=O)N)C(=O)N[C@@H](CC6=CC=CC=C6)C(=O)O)NC(=O)[C@H](CCC(=O)O)NC(=O)[C@H](CO)NC(=O)[C@H](C(C)C)NC(=O)[C@H](CO)N |
| Solubility | Soluble 0.40 mg/mL (water) (Expl.) |
|---|---|
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H332-H351-H361-H372-H373 Details | ||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P264-P270-P271-P280-P301+P317-P304+P340-P317-P318-P319-P330-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Teriparatide acetate is a synthetic peptide composed of the first 34 amino acids of human parathyroid hormone, which represents the biologically active N-terminal fragment. It is classified as an anabolic agent used in the treatment of osteoporosis and other conditions associated with low bone mass. By mimicking the activity of endogenous parathyroid hormone, teriparatide acetate stimulates bone formation through activation of osteoblasts, while also influencing bone remodeling dynamics in favor of increased bone density. The compound was developed to provide a therapeutic option for patients at high risk of fractures, particularly postmenopausal women, men with primary or hypogonadal osteoporosis, and individuals with glucocorticoid-induced osteoporosis. Its design as a peptide fragment allows for selective activation of parathyroid hormone receptors without the complexity and potential adverse effects associated with full-length hormone administration. Teriparatide acetate is administered subcutaneously, typically once daily, due to its peptide nature and rapid degradation if taken orally. Pharmacologically, teriparatide acetate binds to parathyroid hormone type 1 receptors on osteoblasts and osteocytes. This receptor activation triggers intracellular signaling pathways that enhance osteoblast differentiation, proliferation, and activity. Consequently, new bone formation is increased, bone microarchitecture is improved, and bone strength is enhanced. In addition to stimulating bone formation, teriparatide acetate transiently increases bone resorption, but the overall effect favors net bone gain due to the predominant anabolic influence on osteoblasts. Clinical studies have demonstrated that teriparatide acetate significantly increases bone mineral density at key skeletal sites, including the lumbar spine and femoral neck. Treatment with teriparatide acetate has been shown to reduce the incidence of vertebral and non-vertebral fractures, making it an important option for patients at high risk of osteoporosis-related fractures. The therapy is typically limited to a maximum duration of two years, after which transition to antiresorptive therapy is recommended to maintain gains in bone mass. Teriparatide acetate is generally well tolerated, with the most common adverse effects including mild transient hypercalcemia, nausea, dizziness, and leg cramps. Because of its peptide nature, it requires refrigerated storage and proper handling to maintain stability and efficacy. Research continues to explore additional therapeutic applications, including fracture healing and potential use in other skeletal disorders. Overall, teriparatide acetate represents a significant advance in osteoporosis therapy, offering a targeted, bone-building approach that complements traditional antiresorptive treatments. Its development and clinical application have provided important benefits for patients with severe osteoporosis and high fracture risk. References Neer RM, Arnaud CD, Zanchetta JR, et al. (2001) Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. New England Journal of Medicine 344(19) 1434–1441 DOI: 10.1056/NEJM200105103441904 |
| Market Analysis Reports |