Online Database of Chemicals from Around the World
| Shanghai Min-biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.min-biotech.com | |||
 | +86 15190045345 | |||
 | sales@min-biotech.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Cyclic hydrocarbon |
|---|---|
| Name | FR901464 |
| Synonyms | (2S,3Z)-5-{[(2R,3R,5S,6S)-6-{(2E,4E)-5-[(3R,4R,5R,7S)-4,7-dihydroxy-7-methyl-1,6-dioxaspiro[2.5]oct-5-yl]-3-methylpenta-2,4-dien-1-yl}-2,5-dimethyltetrahydro-2H-pyran-3-yl]amino}-5-oxopent-3-en-2-yl acetate |
| Molecular Structure | 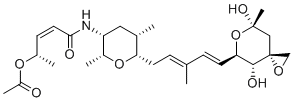 |
| Molecular Formula | C27H41NO8 |
| Molecular Weight | 507.62 |
| CAS Registry Number | 146478-72-0 |
| SMILES | C[C@H]1C[C@H]([C@H](O[C@H]1C/C=C(C)/C=C/[C@@H]2[C@H]([C@@]3(C[C@@](O2)(C)O)CO3)O)C)NC(=O)/C=C[C@H](C)OC(=O)C |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.553, Calc.* |
| Boiling Point | 702.7±60.0 °C (760 mmHg), Calc.* |
| Flash Point | 378.8±32.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS0i7;GHS0i8 Warning Details GHS0i7;GHS0i8 Warning Details |
|---|---|
| Risk Statements | H315-H319-H351 Details |
| Safety Statements | P501-P202-P201-P264-P280-P308+P313-P337+P313-P305+P351+P338-P332+P313-P405 Details |
| SDS | Available |
|
FR901464 was originally isolated from the fermentation broth of Streptomyces roseoflavus, a soil bacterium, by researchers at Fujisawa Pharmaceuticals in Japan. The discovery was serendipitous and resulted from a screening effort to identify novel compounds with anticancer activity. FR901464 belongs to a class of compounds called spliceosome inhibitors, which target the spliceosome, a complex molecular machinery involved in pre-mRNA splicing. The mechanism of action of FR901464 involves binding to the SF3B1 subunit of the spliceosome, disrupting its function and leading to aberrant splicing of mRNA transcripts. Dysregulation of this splicing event leads to the accumulation of unspliced or mis-spliced mRNA species, ultimately triggering cancer cell apoptosis. FR901464 has shown promising therapeutic potential in the treatment of various cancers, including leukemias, lymphomas and solid tumors. Preclinical studies have demonstrated its efficacy in inhibiting tumor growth and inducing apoptosis in cancer cells in vitro and in vivo. Furthermore, FR901464 exhibits selectivity towards cancer cells, sparing normal cells and reducing adverse side effects. In addition to its anticancer effects, FR901464 also shows promise in the field of neuroscience. Studies have shown its potential in regulating neuronal splicing patterns and gene expression, which may provide a new approach to therapeutic intervention in neurological diseases such as Alzheimer's disease, Parkinson's disease and spinal muscular atrophy. References 2024. Synthesis and antiproliferative activity of a tetrahydrofuran analog of FR901464. Bioorganic & Medicinal Chemistry Letters, 104. DOI: 10.1016/j.bmcl.2024.129739 2021. Spliceostatins and Derivatives: Chemical Syntheses and Biological Properties of Potent Splicing Inhibitors. Journal of Natural Products, 84, 5. DOI: 10.1021/acs.jnatprod.1c00100 2001. FR901464: Total Synthesis, Proof of Structure, and Evaluation of Synthetic Analogues. Journal of the American Chemical Society, 123, 38. DOI: 10.1021/ja016615t |
| Market Analysis Reports |