Online Database of Chemicals from Around the World
| Sichuan Zhongbang Technical Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhongbangst.com | |||
 | +86 (830) 258-5019 +86 18982761636 | |||
 | +86 (830) 258-9033 | |||
 | sales@zhongbangst.com sheldon0chen@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Classification | Chemical reagent >> Organic reagent >> Polycyclic compound |
|---|---|
| Name | 1-(Bromomethyl)adamantane |
| Molecular Structure | 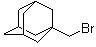 |
| Molecular Formula | C11H17Br |
| Molecular Weight | 229.16 |
| CAS Registry Number | 14651-42-4 |
| EC Number | 690-668-5 |
| SMILES | C1C2CC3CC1CC(C2)(C3)CBr |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 40 - 45 °C (Expl.) |
| Boiling point | 226.5±8.0 °C 760 mmHg (Calc.)* |
| Flash point | 88.1±13.6 °C (Calc.)* |
| Index of refraction | 1.561 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||||||||||||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
1-(Bromomethyl)adamantane is an organic compound with the molecular formula C11H17Br. It is a derivative of adamantane, a highly symmetrical, cage-like tricyclic hydrocarbon known for its exceptional thermal stability and rigid structure. In 1-(bromomethyl)adamantane, a bromomethyl group (-CH2Br) is attached to the 1-position of the adamantane skeleton. Structurally, the molecule consists of the adamantane core, where a single hydrogen atom at the bridgehead (position 1) is replaced by a bromomethyl substituent. This modification imparts reactive functionality to the otherwise inert adamantane framework, allowing 1-(bromomethyl)adamantane to participate in a wide variety of chemical reactions, particularly nucleophilic substitution reactions. The synthesis of 1-(bromomethyl)adamantane typically involves the bromination of 1-methyladamantane (adamantane substituted with a methyl group at the 1-position). The bromination is often performed using N-bromosuccinimide (NBS) in the presence of a radical initiator such as benzoyl peroxide under light or heat. This method favors selective bromination at the methyl group, leading to the desired bromomethyl product with minimal overreaction or side products. Alternative methods may involve direct functionalization of adamantane itself, but these are less commonly employed due to the inherent stability of the adamantane system. 1-(Bromomethyl)adamantane is valuable in organic synthesis as an intermediate for introducing adamantyl groups into various target molecules. The bromine atom serves as a good leaving group, making the compound highly useful for alkylation reactions where the adamantyl moiety can be transferred to nucleophilic partners such as amines, alcohols, or thiols. This approach allows for the preparation of adamantane-containing derivatives with potential applications across multiple fields. In pharmaceutical chemistry, adamantane derivatives are of considerable interest because the adamantyl group imparts unique properties such as enhanced lipophilicity, metabolic stability, and membrane penetration. Notably, compounds like amantadine and memantine, which are adamantane-based drugs, have been successfully developed for antiviral and neuroprotective applications. Therefore, 1-(bromomethyl)adamantane can serve as a precursor for new molecules with similar biological activity profiles. In materials science, adamantane and its derivatives, including 1-(bromomethyl)adamantane, are explored for their ability to introduce rigid and bulky structures into polymers, liquid crystals, and other advanced materials. Their incorporation can improve mechanical strength, thermal stability, and control over molecular packing, leading to materials with desirable optical and electronic properties. The reactivity of 1-(bromomethyl)adamantane is characterized by its behavior as an electrophile. Nucleophilic substitution reactions (both SN1 and SN2 mechanisms depending on conditions) are common, where the bromide is displaced by a nucleophile to form new C–X bonds (where X is the nucleophile). The relatively stable carbocation intermediate that can form during SN1 processes is stabilized by the rigid adamantane structure, favoring efficient reaction pathways under certain conditions. In conclusion, 1-(bromomethyl)adamantane is a highly useful synthetic building block derived from adamantane, providing a reactive bromomethyl handle on a thermally stable, rigid framework. Its applications span organic synthesis, pharmaceutical development, and materials science, reflecting the broad utility of adamantane-based chemistry in modern research and industry. |
| Market Analysis Reports |