Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai CR Corporation Limited | China | |||
|---|---|---|---|---|
 | www.crcorporation.cn | |||
 | +86 (21) 5736-6208 +86 13917420128 | |||
 | +86 (21) 5736-6228 | |||
 | fred.wen@crcorporation.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | 4-Fluoro-2,6-dimethylbromobenzene |
| Synonyms | 4-Bromo-1-fluoro-3,5-dimethylbenzene |
| Molecular Structure | 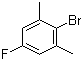 |
| Molecular Formula | C8H8BrF |
| Molecular Weight | 203.05 |
| CAS Registry Number | 14659-58-6 |
| EC Number | 867-360-6 |
| SMILES | CC1=CC(=CC(=C1Br)C)F |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 206.0±35.0 °C 760 mmHg (Calc.)*, 196.5 - 199.3 °C (Expl.) |
| Flash point | 81.1±20.8 °C (Calc.)* |
| Index of refraction | 1.524 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
4-Fluoro-2,6-dimethylbromobenzene is a halogenated aromatic compound derived from the benzene ring system. The molecule is characterized by the presence of two methyl groups at the 2- and 6-positions, a fluorine atom at the 4-position, and a bromine atom directly attached to the aromatic ring. Its molecular formula is C8H8BrF. The study and use of halogenated aromatic compounds such as 4-fluoro-2,6-dimethylbromobenzene are well-established areas in organic and materials chemistry. The synthesis of 4-fluoro-2,6-dimethylbromobenzene can be traced back to research on substituted halobenzenes during the mid-20th century, when chemists were actively developing methods to selectively introduce halogens into aromatic systems. Early methods included the direct bromination of methylated fluorobenzenes under controlled conditions. Later, more refined synthetic techniques, such as halogen exchange reactions and the use of selective catalysts, allowed for higher yields and better positional selectivity. The typical preparation involves bromination of commercially available 4-fluoro-2,6-dimethyltoluene or related intermediates under mild conditions to favor substitution at the desired position without over-bromination or undesired side reactions. In chemical research, 4-fluoro-2,6-dimethylbromobenzene serves as an important intermediate in cross-coupling reactions. The bromine atom, as a good leaving group, enables the compound to undergo palladium-catalyzed Suzuki-Miyaura couplings, allowing the formation of biaryl systems which are valuable motifs in medicinal chemistry and material science. The fluorine substituent, due to its electron-withdrawing nature, can influence the reactivity of the molecule and the properties of the resulting products, such as increasing metabolic stability in drug candidates or modifying the electronic characteristics of materials. The applications of 4-fluoro-2,6-dimethylbromobenzene are mainly centered in synthetic organic chemistry. In the pharmaceutical industry, halogenated aromatics are widely used as building blocks in the synthesis of biologically active molecules. The presence of both methyl groups and fluorine can be used to modulate the pharmacokinetic properties of drugs, affecting factors like solubility, membrane permeability, and binding affinity to biological targets. Although 4-fluoro-2,6-dimethylbromobenzene itself is not a marketed pharmaceutical, it is utilized as an intermediate to construct more complex structures that are biologically relevant. In materials science, the compound is valuable in the design and synthesis of organic electronic materials. Incorporation of fluorine atoms into conjugated systems can enhance the performance of organic light-emitting diodes (OLEDs), organic photovoltaic cells (OPVs), and field-effect transistors (OFETs). The dimethyl substitution provides steric hindrance that can help control the packing and morphology of the material, which are critical factors influencing the electronic and optical properties. Furthermore, 4-fluoro-2,6-dimethylbromobenzene has been used as a starting material for the synthesis of fluorinated polymers and advanced coatings. The introduction of fluorine into polymer backbones improves chemical resistance, lowers surface energy, and increases thermal stability, making the derived materials useful in high-performance applications. The physical properties of 4-fluoro-2,6-dimethylbromobenzene, including its relatively low melting point and moderate boiling point, make it a manageable intermediate in laboratory and industrial settings. Its chemical stability under ambient conditions also contributes to its practicality as a reagent. Research into the safety and handling of brominated aromatics such as 4-fluoro-2,6-dimethylbromobenzene emphasizes caution due to potential toxicity and environmental concerns associated with brominated compounds. Proper handling, storage, and disposal procedures are necessary to minimize risks to health and the environment. In summary, 4-fluoro-2,6-dimethylbromobenzene, a well-defined halogenated aromatic compound, has found notable utility in the fields of organic synthesis, medicinal chemistry, and materials science. Its discovery and development are closely tied to broader advances in the chemistry of halogenated aromatics, highlighting its importance as a synthetic building block in modern chemical research and industry. |
| Market Analysis Reports |