Online Database of Chemicals from Around the World
| Changzhou Jiuwu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuwuchem.com | |||
 | +86 (519) 8512-1086 +86 13775162768 | |||
 | +86 (519) 8512-5369 | |||
 | info@jiuwuchem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2016 | ||||
| Classification | Pharmaceutical intermediate >> OLED material intermediate |
|---|---|
| Name | 9-Vinylcarbazole |
| Synonyms | N-Vinyl carbazole |
| Molecular Structure | 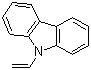 |
| Molecular Formula | C14H11N |
| Molecular Weight | 193.25 |
| CAS Registry Number | 1484-13-5 |
| EC Number | 216-055-0 |
| SMILES | C=CN1C2=CC=CC=C2C3=CC=CC=C31 |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 60-65 °C (Expl.) |
| Index of Refraction | 1.610, Calc.* |
| Boiling Point | 328.5±15.0 °C (760 mmHg), Calc.*, 154-155 °C (3 mmHg) (Expl.) |
| Flash Point | 152.5±20.4 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |     GHS06;GHS07;GHS08;GHS09 Danger Details GHS06;GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H300-H312-H315-H317-H341-H400-H410 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P264-P270-P272-P273-P280-P301+P316-P302+P352-P317-P318-P321-P330-P332+P317-P333+P317-P362+P364-P391-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
9-Vinylcarbazole is a pivotal compound in materials science and organic chemistry, known for its aromatic carbazole structure and a reactive vinyl group. These features enable its use in producing advanced polymers and materials, particularly in the optoelectronics and photonics sectors. The discovery of 9-vinylcarbazole emerged during investigations into carbazole derivatives in the mid-20th century. Carbazole itself is naturally occurring, and its derivatives were initially explored for their biological activities. Modifications such as the introduction of a vinyl group expanded its utility, particularly in materials chemistry. 9-Vinylcarbazole is typically synthesized via alkylation of carbazole, often using acetylene derivatives or halogenated alkyl precursors under basic or catalytic conditions. One of the most notable applications of 9-vinylcarbazole is in the production of poly(N-vinylcarbazole) (PVK), a polymer with significant applications in electronics. PVK exhibits excellent photoconductive and charge-transport properties, making it ideal for use in organic light-emitting diodes (OLEDs), photovoltaic devices, and photoreceptors in xerography. Its ability to function as a hole-transporting material has also driven innovation in optoelectronics. In addition to its use in electronic applications, 9-vinylcarbazole serves as a precursor for functionalized polymers and copolymers. By co-polymerizing with other monomers, it enables the development of materials with tailored properties for specific industrial applications, such as coatings, adhesives, and conductive films. The pharmaceutical field has also explored carbazole derivatives for their potential therapeutic properties, though the vinyl group in 9-vinylcarbazole limits its direct biological use due to reactivity. However, its derivatives and copolymers have shown promise in drug delivery systems and biocompatible materials. Research on 9-vinylcarbazole continues to advance, focusing on enhancing its polymerization techniques, optimizing its electronic properties, and expanding its applications in renewable energy and sustainable materials. Developments in nanotechnology have also leveraged its structure for creating advanced nanocomposites and hybrid materials. References 2024. Microfluidic vs. batch synthesis of fluorescent poly(GMA-co-EGDMA) micro/nanoparticles for biomedical applications. Emergent Materials, 10, 5. DOI: 10.1007/s42247-024-00840-9 2024. Palladium-catalyzed vinylation of indoles with vinyl ethers via deoxygenative coupling. Science China Chemistry, 67, 9. DOI: 10.1007/s11426-024-2184-x 1964. Über ein Dimeres des N-Vinylcarbazols. Monatshefte fur Chemie und verwandte Teile anderer Wissenschaften, 95, 6. DOI: 10.1007/bf00908815 |
| Market Analysis Reports |