Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Apexbio Technology LLC | USA | |||
|---|---|---|---|---|
 | www.apexbt.com | |||
 | +1 (832) 696-8203 | |||
 | +1 (855) 527-3928 | |||
 | info@apexbt.com | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Guangzhou Green Cross Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.m-pharma.com.cn | |||
 | +86 (20) 8222-0238 | |||
 | wangsy@m-pharma.com.cn | |||
| Chemical manufacturer since 1991 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Ramidus AB | Sweden | |||
|---|---|---|---|---|
 | www.ramidus.se | |||
 | +46 (46) 286-2850 | |||
 | +46 (46) 286-2851 | |||
 | info@ramidus.se | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Glycine derivatives |
|---|---|
| Name | Sivelestat sodium |
| Synonyms | N-[2-[[[4-(2,2-Dimethyl-1-oxopropoxy)phenyl]sulfonyl]amino]benzoyl]-(S)-glycine monosodium salt |
| Molecular Structure | 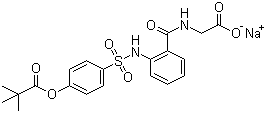 |
| Molecular Formula | C20H21N2NaO7S |
| Molecular Weight | 456.44 |
| Protein Sequence | XG |
| CAS Registry Number | 150374-95-1 |
| SMILES | CC(C)(C)C(=O)OC1=CC=C(C=C1)S(=O)(=O)NC2=CC=CC=C2C(=O)NCC(=O)[O-].[Na+] |
| Solubility | Soluble 50 mM in DMSO (Expl.) |
|---|---|
|
Sivelestat sodium is the sodium salt form of sivelestat, a synthetic low-molecular-weight inhibitor of human neutrophil elastase (HNE). Neutrophil elastase is a serine protease enzyme released by activated neutrophils during inflammatory responses. While this enzyme plays a role in the destruction of pathogens and damaged tissues, excessive activity of neutrophil elastase can contribute to tissue injury and inflammation, particularly in the lungs. Sivelestat sodium was developed to counteract these harmful effects by selectively inhibiting neutrophil elastase. The discovery of sivelestat sodium emerged from research focused on identifying compounds that could inhibit neutrophil elastase with high specificity and efficacy. Neutrophil elastase was implicated in the pathology of acute lung injury (ALI) and acute respiratory distress syndrome (ARDS), which involve inflammatory damage to the pulmonary tissues. Sivelestat sodium was developed by Ono Pharmaceutical Co., Ltd., and introduced in Japan in the early 2000s. It is marketed under the brand name Elaspol. Sivelestat sodium is administered intravenously and is used clinically in Japan and some other Asian countries for the treatment of ALI associated with systemic inflammatory response syndrome (SIRS). It works by binding to the active site of neutrophil elastase, thereby inhibiting its proteolytic activity. By limiting the enzyme's action, sivelestat sodium helps reduce inflammatory damage to alveolar structures, decrease capillary permeability, and improve pulmonary function in affected patients. Clinical studies in Japan have reported improvements in pulmonary parameters, such as oxygenation indices, in patients with ALI and SIRS treated with sivelestat sodium. However, the overall impact on mortality rates and long-term outcomes has been a subject of debate. As a result, the use of sivelestat sodium remains limited to specific indications, and it has not been widely approved outside of Asia. In addition to ALI and ARDS, sivelestat sodium has been investigated for potential benefits in other conditions involving neutrophil-mediated tissue injury. These include ischemia-reperfusion injury, pancreatitis, and certain cardiovascular and gastrointestinal disorders. However, such applications remain under study, and sivelestat sodium is not approved for these uses. Pharmacokinetically, sivelestat sodium is rapidly distributed in the body following intravenous administration, and it is metabolized primarily in the liver. The drug has a short half-life and requires continuous infusion or repeated dosing to maintain therapeutic levels. It is generally well tolerated, but possible side effects include liver function abnormalities, gastrointestinal symptoms, and allergic reactions. Monitoring of liver enzymes and clinical status is advised during therapy. The development of sivelestat sodium highlights the ongoing interest in targeted anti-inflammatory therapies that can modulate specific components of the immune response. Neutrophil elastase inhibitors represent one approach among many that seek to reduce tissue damage caused by excessive or dysregulated inflammation. In summary, sivelestat sodium is a selective neutrophil elastase inhibitor developed for the treatment of acute lung injury associated with systemic inflammation. Its medical use is primarily centered in Japan and a few other regions, with clinical application supported by its ability to reduce inflammation and preserve lung function. While further research is ongoing, the compound remains a representative example of enzyme-targeted drug development in the context of inflammatory diseases. References 1991. ONO-5046, a novel inhibitor of human neutrophil elastase. Biochemical and Biophysical Research Communications, 177(2). DOI: 10.1016/0006-291x(91)91862-7 2023. Synthesis of Related Substances of Sivelestat Sodium. Russian Journal of General Chemistry, 93(12). DOI: 10.1134/s1070363223120162 2024. Possible pharmacological targets and mechanisms of sivelestat in protecting acute lung injury. Computers in Biology and Medicine, 170. DOI: 10.1016/j.compbiomed.2024.108080 |
| Market Analysis Reports |