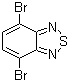
4,7-Dibromo-2,1,3-benzothiadiazole is a compound with the chemical formula C₆H₂Br₂N₂S that has attracted great interest in both scientific research and industrial applications since its discovery. Characterized by a benzothiadiazole core and bromine substituents, this compound plays a key role in the development of advanced materials, especially in the field of organic electronics.
The discovery of 4,7-dibromo-2,1,3-benzothiadiazole dates back to the mid-20th century, when chemists were exploring the unique electronic properties of heterocyclic compounds. The incorporation of bromine atoms into the benzothiadiazole structure enhances its chemical reactivity, making it a valuable building block for a variety of synthetic applications. The unique properties of this compound stem from the electron-withdrawing nature of the bromine atom and the electron-rich benzothiadiazole core, which together influence the electronic properties of the materials it helps create.
One of the most notable applications of 4,7-dibromo-2,1,3-benzothiadiazole is in the field of organic photovoltaics (OPVs). Organic solar cells are a flexible, lightweight alternative to traditional silicon-based solar cells that rely on the precise engineering of organic molecules to efficiently harvest sunlight and convert it into electricity. 4,7-Dibromo-2,1,3-benzothiadiazole is a key ingredient in the design of donor-acceptor copolymers that are essential for the active layer of these solar cells. The compound is integral in improving OPV efficiency due to its ability to tune electronic properties and enhance charge transport.
In addition to its role in the solar energy sector, 4,7-Dibromo-2,1,3-benzothiadiazole plays a vital role in the development of organic light-emitting diodes (OLEDs). OLEDs are used in a variety of display technologies, including televisions, smartphones, and other digital screens. The compound combined with the emissive layer of OLEDs helps achieve high brightness and color purity, which are essential for vivid and energy-efficient displays. 4,7-Dibromo-2,1,3-benzothiadiazole's versatility in fine-tuning the photophysical properties of organic materials underpins its widespread use in this field.
Additionally, the compound plays an important role in the synthesis of organic field-effect transistors (OFETs), which are key components of flexible electronic devices. OFETs benefit from the compound’s ability to enhance charge carrier mobility, which improves the performance and reliability of these devices. This makes 4,7-dibromo-2,1,3-benzothiadiazole a cornerstone in the development of flexible and wearable electronics, which are becoming increasingly common in modern technology.
Despite its many advantages, 4,7-dibromo-2,1,3-benzothiadiazole requires careful consideration of safety protocols when handling and synthesizing it due to the potential hazards of bromine compounds. Proper laboratory practices and environmental regulations are essential to mitigate any risks during use and disposal.
References
2021. Synthesis by direct arylation reaction of photovoltaic D-μ-A polymers based on fluorene-thiophene- fragment and fluorinated benzothiadiazole derivatives. Journal of Materials Science: Materials in Electronics, 30, 13739-13747.
DOI: 10.1007/s10854-019-01744-9
2022. Derivatives of diphenylamine and benzothiadiazole in optoelectronic applications: a review. Journal of Polymer Research, 29, 347.
DOI: 10.1007/s10965-022-03266-1
2020. Synthesis of benzothiadiazole-9,9-bis(perfluorohexylethyl propionate) fluorene copolymers with enhanced photoluminance stability. Journal of Materials Science: Materials in Electronics, 32, 24567-24576.
DOI: 10.1007/s10854-021-06327-1
|





























































 GHS06 Danger Details
GHS06 Danger Details