Online Database of Chemicals from Around the World
| MolScanner | Singapore | |||
|---|---|---|---|---|
 | www.molscanner.com | |||
 | +86 18621675448 | |||
 | marketing@molscanner.com | |||
 | WhatsApp:9896 7603 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Carboxylic acid |
|---|---|
| Name | 3-Bromo-6-fluoro-2-hydroxybenzoic acid |
| Molecular Structure | 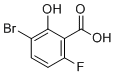 |
| Molecular Formula | C7H4BrFO3 |
| Molecular Weight | 235.01 |
| CAS Registry Number | 1538235-22-1 |
| SMILES | C1=CC(=C(C(=C1F)C(=O)O)O)Br |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P264-P270-P271-P280-P302+P352-P304+P340-P321-P330-P362+P364-P405-P501 Details |
| SDS | Available |
|
3-Bromo-6-fluoro-2-hydroxybenzoic acid is a halogenated derivative of salicylic acid, featuring a benzene ring substituted with a hydroxyl group at the 2-position (ortho to the carboxylic acid), a bromine atom at the 3-position, a fluorine atom at the 6-position, and a carboxylic acid group at the 1-position. This molecular structure places it within the class of halogenated hydroxybenzoic acids, a group of compounds often valued for their chemical versatility and biological relevance. The synthesis of 3-bromo-6-fluoro-2-hydroxybenzoic acid typically proceeds from appropriately substituted benzoic acid derivatives through selective halogenation reactions. The introduction of bromine and fluorine atoms at specific positions on the aromatic ring can be achieved using controlled electrophilic aromatic substitution or via directed ortho-metalation followed by halogenation. Alternatively, cross-coupling strategies, such as halogen exchange or transition-metal-catalyzed halogenation, can be used to prepare such compounds with high regioselectivity. This compound's structure is defined by several key functional groups: the phenolic hydroxyl group, the carboxylic acid, and two electron-withdrawing halogens. The combination of these groups contributes to the compound's acidity, reactivity, and solubility in polar solvents. The presence of both bromine and fluorine atoms influences the molecule’s electron distribution and can modulate interactions in both chemical and biological systems. 3-Bromo-6-fluoro-2-hydroxybenzoic acid is used as an intermediate in the synthesis of more complex molecules, particularly in pharmaceutical and agrochemical research. The hydroxyl and carboxylic acid functionalities make it suitable for further derivatization into esters, amides, and ethers, while the halogen atoms provide sites for palladium-catalyzed cross-coupling reactions, such as Suzuki or Sonogashira couplings. These transformations are fundamental in modern synthetic organic chemistry, especially for constructing diverse aromatic frameworks. Compounds related to halogenated salicylic acids have been studied for a range of biological activities, including antimicrobial, anti-inflammatory, and herbicidal properties. While 3-bromo-6-fluoro-2-hydroxybenzoic acid itself is not a widely used drug or agrochemical, its structural motif appears in analogues with known biological relevance. It serves as a valuable scaffold for structure–activity relationship (SAR) studies, enabling the exploration of the influence of halogen positioning and substitution on biological function. In material science, hydroxybenzoic acid derivatives also find occasional use in coordination chemistry and supramolecular systems due to their ability to act as ligands, forming complexes with metal ions through the hydroxyl and carboxylic acid groups. The addition of halogen atoms further modulates these properties and can affect solid-state packing and thermal behavior. In summary, 3-bromo-6-fluoro-2-hydroxybenzoic acid is a synthetically accessible, functionally rich aromatic compound used primarily as an intermediate in organic synthesis. Its combination of reactive sites and modifiable substituents makes it a useful building block in the development of pharmaceuticals, agrochemicals, and advanced materials. |
| Market Analysis Reports |