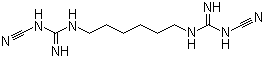
1,6-Bis(cyano-guanidino)hexane is an organic compound characterized by the presence of two cyano-guanidino functional groups attached at the terminal positions of a hexane (six-carbon) aliphatic chain. The molecular structure consists of a linear hexamethylene backbone substituted at both ends with cyano-guanidino moieties, which contain nitrogen-rich groups notable for their strong basicity and potential for multiple hydrogen bonding interactions.
The synthesis of 1,6-bis(cyano-guanidino)hexane typically involves multi-step organic reactions starting from hexanediamine or related precursors. The terminal amine groups of hexanediamine undergo functionalization to introduce the cyano-guanidino groups, often through reactions with cyanamide derivatives or guanidinylating agents under controlled conditions. These transformations result in the formation of stable guanidine functionalities bearing cyano substituents, conferring distinctive electronic and coordination properties.
This compound is of interest primarily in the field of coordination chemistry and as a ligand due to the presence of guanidine groups, which are strong Lewis bases capable of coordinating to transition metals through nitrogen atoms. The cyano substituents adjacent to the guanidino groups enhance the electron-withdrawing character, potentially modulating the binding affinity and electronic effects in coordination complexes. Such features make 1,6-bis(cyano-guanidino)hexane a candidate ligand for catalysis, metal-organic frameworks, and supramolecular assemblies.
Additionally, 1,6-bis(cyano-guanidino)hexane may be investigated for its biological and pharmaceutical relevance, as guanidine derivatives are known for their bioactivity, including enzyme inhibition and receptor binding. The presence of the cyano groups may influence the compound’s solubility, cell permeability, and interaction with biological macromolecules.
In materials science, the nitrogen-rich nature and potential for strong hydrogen bonding of this compound make it useful as a building block in the design of polymeric materials, ionic liquids, or as a component in molecular recognition systems. Its bifunctional nature allows for cross-linking or bridging interactions in complex architectures.
Handling of 1,6-bis(cyano-guanidino)hexane requires appropriate safety measures as guanidine derivatives can be irritants and may be toxic if ingested or inhaled. The compound should be stored in a dry, well-ventilated environment away from strong oxidizing agents and acids.
In summary, 1,6-bis(cyano-guanidino)hexane is a bifunctional organic compound featuring cyano-guanidino groups at both ends of a hexane chain. It serves as a potentially valuable ligand in coordination chemistry, with applications spanning catalysis, materials science, and biological research, due to its strong basicity, hydrogen bonding ability, and electronic properties.
References
2023. Quantitation of guanidine derivatives as representative persistent and mobile organic compounds in water: method development. Analytical and Bioanalytical Chemistry, 415(11).
DOI: 10.1007/s00216-023-04613-x
2006. Biodegradability of end-groups of the biocide polyhexamethylene biguanide (PHMB) assessed using model compounds. Journal of Industrial Microbiology and Biotechnology, 33(7).
DOI: 10.1007/s10295-006-0103-6
|



































 GHS07 Warning Details
GHS07 Warning Details