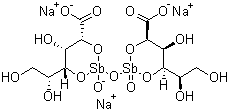
Sodium stibogluconate, a compound with significant medical applications, was first introduced in the mid-20th century. It is a derivative of stibogluconate and belongs to a class of antimonial drugs used primarily in the treatment of parasitic diseases.
The primary application of Sodium stibogluconate is in the treatment of leishmaniasis, a parasitic disease caused by protozoa of the genus Leishmania. Leishmaniasis can manifest in several forms, including cutaneous, mucocutaneous, and visceral leishmaniasis, which can be severe and life-threatening if left untreated. Sodium stibogluconate acts as an antileishmanial agent by interfering with the metabolism of the parasite, ultimately leading to its destruction. The compound is administered intravenously or intramuscularly, and its effectiveness in treating leishmaniasis has made it a key drug in regions where the disease is endemic.
In addition to its use in leishmaniasis treatment, Sodium stibogluconate has applications in the management of other parasitic infections. It has been explored for its potential efficacy against various protozoan parasites, contributing to the broader field of parasitic disease management. Its role in chemotherapy and parasitology highlights its importance in both clinical and research settings.
The discovery of Sodium stibogluconate marked a significant advancement in the treatment of parasitic diseases, offering an effective therapeutic option where other treatments were limited. Its development was driven by the need for targeted treatments for diseases prevalent in tropical and subtropical regions.
Despite its efficacy, the use of Sodium stibogluconate must be carefully managed due to potential side effects and the need for appropriate dosing to minimize toxicity. Ongoing research continues to evaluate its safety profile and explore new formulations to enhance its therapeutic potential.
References
1945. The chemotherapy of experimental leishmaniasis II. A dose response curve for the activity of sodium stibogluconate. Transactions of the Royal Society of Tropical Medicine and Hygiene, 39, 117-126.
DOI: 10.1016/0035-9203(45)90004-6
2024. Repurposing sodium stibogluconate as an uracil DNA glycosylase inhibitor against prostate cancer using a time-resolved oligonucleotide-based drug screening platform. Bioorganic Chemistry, 144, 107176.
DOI: 10.1016/j.bioorg.2024.107176
|




























 GHS07;GHS09 Warning Details
GHS07;GHS09 Warning Details