Online Database of Chemicals from Around the World
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Ryan Scientific, Inc. | USA | |||
|---|---|---|---|---|
 | www.ryansci.com | |||
 | +1 (843)-884-4911 | |||
 | +1 (843) 884-5568 | |||
 | sales@ryansci.com | |||
| Chemical manufacturer | ||||
| SynChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.synchem.com | |||
 | +1 (847) 298-2436 ex 230 | |||
 | +1 (847) 298-2439 | |||
 | sales@synchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Piperazine |
|---|---|
| Name | 1-Methyl-4-(4-nitrophenyl)piperazine |
| Molecular Structure | 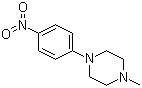 |
| Molecular Formula | C11H15N3O2 |
| Molecular Weight | 221.26 |
| CAS Registry Number | 16155-03-6 |
| SMILES | CN1CCN(CC1)C2=CC=C(C=C2)[N+](=O)[O-] |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
1-Methyl-4-(4-nitrophenyl)piperazine is an important compound in the field of medicinal chemistry, primarily known for its role as a building block in the synthesis of various pharmaceuticals and bioactive molecules. Its discovery is linked to the ongoing exploration of piperazine derivatives, a class of compounds that have been extensively studied due to their diverse pharmacological activities. The structure of 1-Methyl-4-(4-nitrophenyl)piperazine is characterized by a piperazine ring substituted with a methyl group at the nitrogen atom and a 4-nitrophenyl group attached to one of the ring's carbon atoms. This combination of functional groups imparts specific chemical properties to the molecule, making it a versatile intermediate in synthetic organic chemistry. One of the key applications of 1-Methyl-4-(4-nitrophenyl)piperazine is in the development of pharmaceutical agents, particularly those targeting the central nervous system (CNS). Piperazine derivatives, including this compound, have been investigated for their potential as anxiolytics, antidepressants, and antipsychotics. The nitrophenyl group in its structure plays a crucial role in modulating the biological activity of the molecule, contributing to its binding affinity and selectivity for various receptors in the brain. In addition to its role in CNS drug development, 1-Methyl-4-(4-nitrophenyl)piperazine has found use in the synthesis of compounds with antimicrobial and antiparasitic activities. The piperazine core structure is a common motif in many drugs used to treat infections and parasitic diseases, and the nitrophenyl substitution can enhance the potency and spectrum of activity of these agents. The compound is also valuable as a precursor in the synthesis of more complex molecules. The presence of the nitro group provides a site for further chemical modification, such as reduction to an amine or substitution reactions, which can lead to the creation of a wide variety of derivatives with potential therapeutic applications. Moreover, 1-Methyl-4-(4-nitrophenyl)piperazine has been utilized in the preparation of chemical probes and diagnostic tools. These probes are used in biological research to investigate the function of specific proteins or enzymes, particularly those involved in neurological and infectious diseases. The ability to selectively modify the nitrophenyl group allows researchers to tailor the properties of the probes for specific applications, enhancing their utility in experimental settings. The discovery of 1-Methyl-4-(4-nitrophenyl)piperazine and its subsequent development into a key intermediate in pharmaceutical synthesis highlights the importance of piperazine derivatives in modern medicinal chemistry. As research continues, the compound's versatility and efficacy in drug development and biological research are likely to expand, contributing to the advancement of new therapies and diagnostic tools. References 1973. Synthesis of the antitubercular preparation butomelide. Pharmaceutical Chemistry Journal, 7(4), 235-238. DOI: 10.1007/bf00771516 2021. C-H Bond Functionalization of Amines: A Graphical Overview of Diverse Methods. SynOpen (2017), 5(3), 210-230. DOI: 10.1055/s-0040-1706051 2020. Discovery of 5-(4-methylpiperazin-1-yl)-2-nitroaniline derivatives as a new class of SIRT6 inhibitors. Bioorganic & Medicinal Chemistry Letters, 30(16), 127215. DOI: 10.1016/j.bmcl.2020.127215 |
| Market Analysis Reports |