Online Database of Chemicals from Around the World
| Shandong Benrite New Chemical Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.benritechem.com | |||
 | +86 (0533) 286-6658 | |||
 | sales@benritechem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: benritechem | |||
 | WhatsApp:+8613021512891 | |||
| Chemical distributor since 2024 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Chemical reagent >> Organic reagent >> Sulfonate / sulfinate |
|---|---|
| Name | Propargyl mesylate |
| Synonyms | prop-2-ynyl methanesulfonate |
| Molecular Structure | 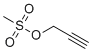 |
| Molecular Formula | C4H6O3S |
| Molecular Weight | 134.15 |
| CAS Registry Number | 16156-58-4 |
| EC Number | 808-222-7 |
| SMILES | CS(=O)(=O)OCC#C |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.459, Calc.* |
| Boiling Point | 246.7±23.0 °C (760 mmHg), Calc.* |
| Flash Point | 103.0±22.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS05;GHS06;GHS07 Danger Details GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H314-H315-H319 Details | ||||||||||||||||
| Safety Statements | P260-P264-P264+P265-P270-P280-P301+P316-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P351+P338-P305+P354+P338-P316-P321-P330-P332+P317-P337+P317-P362+P364-P363-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| Transport Information | UN 2810 | ||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Propargyl mesylate is an important alkylating agent widely used in synthetic organic chemistry. Structurally, it consists of a propargyl group (HC≡C-CH2-) linked to a mesylate group (-SO2CH3), which makes it highly reactive in nucleophilic substitution reactions. This compound plays a significant role in the preparation of complex molecules, including pharmaceuticals, natural products, and materials. Propargyl mesylate was first identified in the mid-20th century during the exploration of propargyl compounds for their versatility in organic synthesis. Chemists recognized that the introduction of a mesylate group on a propargyl backbone provided an efficient leaving group, enhancing its reactivity toward nucleophiles. This discovery led to numerous applications in synthetic strategies where alkynes serve as key intermediates. The synthesis of propargyl mesylate is straightforward and typically involves the mesylation of propargyl alcohol. This process uses methanesulfonyl chloride (MsCl) in the presence of a base such as triethylamine or pyridine to facilitate the substitution of the hydroxyl group with a mesylate group. The reaction conditions are mild, and the product can be isolated with high yields, making it an attractive method for large-scale synthesis. Propargyl mesylate is highly valuable in the formation of carbon-carbon bonds through nucleophilic substitution reactions. It serves as a versatile building block in the synthesis of heterocycles, pharmaceuticals, and other bioactive molecules. For instance, it is commonly used in the preparation of alkynyl-substituted heterocycles and macrocycles. In addition, propargyl mesylate is employed in click chemistry, particularly in copper-catalyzed azide-alkyne cycloaddition (CuAAC), which is widely used for constructing 1,2,3-triazoles. This compound also finds applications in the synthesis of advanced materials, such as polymers and dendrimers, where the incorporation of propargyl units introduces functional sites for further modification. Its role in medicinal chemistry includes the development of enzyme inhibitors, antiviral agents, and other therapeutic compounds, where the alkyne moiety provides a handle for structural diversification. Research continues to explore the potential of propargyl mesylate in innovative synthetic methodologies and the development of new materials. Its ease of synthesis, stability, and high reactivity ensure its enduring importance in organic chemistry. References 2004. Total Synthesis of Amphidinolide X. Journal of the American Chemical Society, 126(46), 15012-15013. DOI: 10.1021/ja044130+ 2021. Synthesis of five-membered nitrogen-containing heterocycles using copper. Journal of the Iranian Chemical Society, 18(12), 3153-3165. DOI: 10.1007/s13738-021-02350-5 2010. Efficient Total Synthesis of (S)‐14‐Azacamptothecin. Chemistry - An Asian Journal, 5(6), 1361-1369. DOI: 10.1002/asia.201000073 |
| Market Analysis Reports |