Online Database of Chemicals from Around the World
| Shanghai Rui Yun Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rychemical.com.cn | |||
 | +86 (21) 6726-7633 | |||
 | +86 (21) 6726-7655 | |||
 | sales@rychemical.com.cn | |||
 | WeChat: 13917251563 | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2009 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Carboxylic acid |
|---|---|
| Name | 4-Bromo-1-naphthoic acid |
| Molecular Structure | 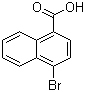 |
| Molecular Formula | C11H7BrO2 |
| Molecular Weight | 251.08 |
| CAS Registry Number | 16650-55-8 |
| EC Number | 640-764-8 |
| SMILES | C1=CC=C2C(=C1)C(=CC=C2Br)C(=O)O |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H319-H400-H410 Details | ||||||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P273-P280-P301+P317-P305+P351+P338-P330-P337+P317-P391-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
4-Bromo-1-naphthoic acid is a chemical compound characterized by its unique structure, which incorporates both a bromine atom and a naphthoic acid moiety. This compound is notable for its utility in various fields of chemical research and industry due to its distinct chemical properties and reactivity. The discovery of 4-bromo-1-naphthoic acid dates back to studies exploring the functionalization of naphthoic acid derivatives. The introduction of a bromine atom into the naphthoic acid structure was a strategic modification aimed at enhancing the compound's reactivity and expanding its applications. The synthesis of this compound typically involves bromination of naphthoic acid under controlled conditions, resulting in the formation of the 4-bromo derivative. In organic synthesis, 4-bromo-1-naphthoic acid serves as an important intermediate. The presence of the bromine atom makes it a valuable reagent for further chemical transformations. Bromine is a potent leaving group, facilitating various substitution and coupling reactions. This property enables chemists to use 4-bromo-1-naphthoic acid in the synthesis of more complex molecules, including pharmaceuticals and materials. One of the primary applications of 4-bromo-1-naphthoic acid is in the development of pharmaceuticals. The compound's structure allows for the introduction of additional functional groups, which can be utilized to create novel drug candidates. For example, the bromine atom can participate in cross-coupling reactions with other organic substrates, leading to the formation of biologically active molecules. Researchers often explore its potential in the synthesis of therapeutic agents with enhanced efficacy or selectivity. In materials science, 4-bromo-1-naphthoic acid is used to modify and functionalize materials. The brominated naphthoic acid can be incorporated into polymer matrices, where it can influence the material's properties, such as its solubility, thermal stability, and reactivity. These modifications can be useful in designing advanced materials with tailored characteristics for specific applications. Furthermore, 4-bromo-1-naphthoic acid is employed in the production of dyes and pigments. The naphthoic acid framework provides a basis for the synthesis of colorants with desirable properties. By altering the naphthoic acid structure through bromination, researchers can develop dyes with improved stability and color performance for various industrial applications. Overall, 4-bromo-1-naphthoic acid is a versatile chemical compound with significant applications in organic synthesis, pharmaceuticals, materials science, and dye production. Its unique combination of bromine and naphthoic acid functionalities makes it a valuable tool for researchers and industry professionals seeking to explore new chemical transformations and applications. References 2022. Synthesis of Verinurad. Synfacts. DOI: 10.1055/s-0041-1738393 2007. Response of the bioluminescent bioreporter Pseudomonas fluorescens HK44 to analogs of naphthalene and salicylic acid. Folia Microbiologica. DOI: https://pubmed.ncbi.nlm.nih.gov/17571789 2000. Perylene derivatives formation in reaction of 3-bromobenzanthrone and 4-bromonaphthalic acid derivatives with a reduction system NiCl2-2,2′bipyridyl (or 1,10-phenathroline)-Zn. Russian Journal of Electrochemistry. DOI: 10.1007/bf02757446 |
| Market Analysis Reports |