Online Database of Chemicals from Around the World
| Chizhou Dongsheng Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dspharmchem.com | |||
 | +86 (576) 8429-2571 | |||
 | +86 (576) 8429-2560 | |||
 | ksz@dspharmchem.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | API >> Inhibitor drug |
|---|---|
| Name | (3-Fluoro-4-morpholin-4-ylphenyl)carbamic acid benzyl ester |
| Synonyms | benzyl N-(3-fluoro-4-morpholin-4-ylphenyl)carbamate |
| Molecular Structure | 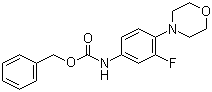 |
| Molecular Formula | C18H19FN2O3 |
| Molecular Weight | 330.35 |
| CAS Registry Number | 168828-81-7 |
| EC Number | 605-533-8 |
| SMILES | C1COCCN1C2=C(C=C(C=C2)NC(=O)OCC3=CC=CC=C3)F |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
(3-Fluoro-4-morpholin-4-ylphenyl)carbamic acid benzyl ester is a synthetic organic compound that belongs to the class of carbamates. Carbamates are widely used in various fields, including medicinal chemistry and materials science, due to their versatile chemical properties and ability to form stable bonds with biological molecules. The discovery of this specific compound can be attributed to research efforts in the field of drug development, particularly aimed at modifying molecular structures to enhance pharmacological activity and improve drug-like properties. The structure of (3-Fluoro-4-morpholin-4-ylphenyl)carbamic acid benzyl ester contains several functional groups that contribute to its chemical behavior and potential applications. The fluorine atom, attached to the phenyl ring, is known to influence the compound's lipophilicity and metabolic stability. Fluorine substitution is commonly employed in medicinal chemistry to increase the bioavailability and potency of pharmaceutical agents. The morpholine group, a six-membered ring containing oxygen and nitrogen, enhances the molecule's solubility in water and may impact its interaction with biological targets, particularly receptors and enzymes. The benzyl ester group serves as a protecting group, which can be hydrolyzed in vivo, releasing the active compound in a controlled manner. Applications of this compound are primarily focused on its potential in medicinal chemistry. Due to its structural characteristics, (3-Fluoro-4-morpholin-4-ylphenyl)carbamic acid benzyl ester may serve as an intermediate or prodrug in the development of pharmaceuticals targeting specific diseases, such as cancer or inflammatory disorders. The presence of the fluorine atom could improve the compound’s binding affinity to certain enzymes or receptors, while the carbamate group may provide stability and resistance to enzymatic degradation. Researchers have explored similar carbamate derivatives for their activity as enzyme inhibitors, particularly in the case of acetylcholinesterase inhibitors used in the treatment of neurodegenerative diseases such as Alzheimer’s. The introduction of a morpholine moiety enhances the pharmacokinetic profile, making these compounds more soluble and easier to deliver through various routes of administration. In addition to its medicinal applications, the compound may have uses in materials science and polymer chemistry. Carbamate groups are known to form stable linkages, making such compounds useful in the synthesis of polymers with specialized properties. The ester group in (3-Fluoro-4-morpholin-4-ylphenyl)carbamic acid benzyl ester could participate in polymerization reactions, leading to materials with potential applications in coatings, adhesives, and biomedical devices. The continued research into the applications of carbamate derivatives like this compound highlights the importance of synthetic organic chemistry in advancing drug development and materials science. By modifying molecular structures with functional groups such as fluorine and morpholine, chemists can fine-tune the properties of these compounds for specific applications, making them valuable tools in both medicine and industry. References 2003. Linezolid. Pharmaceutical Substances. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-12-0041 |
| Market Analysis Reports |