Online Database of Chemicals from Around the World
| Chizhou Dongsheng Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dspharmchem.com | |||
 | +86 (576) 8429-2571 | |||
 | +86 (576) 8429-2560 | |||
 | ksz@dspharmchem.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Organic raw materials >> Amino compound >> Amide compound |
|---|---|
| Name | (S)-5-(Aminomethyl)-3-(3-fluoro-4-morpholinophenyl)oxazolidin-2-one |
| Synonyms | (5S)-5-(Aminomethyl)-3-(3-fluoro-4-morpholinophenyl)-1,3-oxazolidin-2-one; (S)-(-)-5-Aminomethyl-3-(3-fluoro-4-morpholinophenyl)-1,3-oxazolidin-2-one; (S)-N-[[3-[3-Fluoro-4-(4-morpholinyl)phenyl]-2-oxo-5-oxazolidinyl]methyl]amine |
| Molecular Structure | 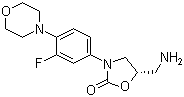 |
| Molecular Formula | C14H18FN3O3 |
| Molecular Weight | 295.31 |
| CAS Registry Number | 168828-90-8 |
| SMILES | C1COCCN1C2=C(C=C(C=C2)N3C[C@@H](OC3=O)CN)F |
| Density | 1.319 |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H332-H335 Details |
| Safety Statements | P261-P280-P305+P351+P338 Details |
| SDS | Available |
|
(S)-5-(Aminomethyl)-3-(3-fluoro-4-morpholinophenyl)oxazolidin-2-one is a compound of considerable interest in the field of medicinal chemistry, particularly for its potential use in treating bacterial infections. This oxazolidinone derivative is part of a class of antibiotics known for their ability to inhibit bacterial protein synthesis, making it a candidate for addressing the growing challenge of antibiotic resistance. The discovery of (S)-5-(Aminomethyl)-3-(3-fluoro-4-morpholinophenyl)oxazolidin-2-one builds on the foundation of existing oxazolidinones, such as linezolid, which has been effective against Gram-positive bacteria. The specific structure of this compound includes several functional groups that enhance its antimicrobial properties. The oxazolidinone ring, which is the core structure of the molecule, plays a crucial role in its ability to bind to bacterial ribosomes and inhibit protein synthesis. The (S)-stereochemistry of the compound is significant, as the chirality of the oxazolidinone ring can impact its biological activity and interaction with bacterial targets. The aminomethyl group at position 5 is essential for the compound's activity, potentially affecting its binding affinity and stability. The presence of a fluorine atom and a morpholine group on the phenyl ring contributes to the molecule's pharmacokinetic properties. Fluorine is known for enhancing metabolic stability and bioavailability, while the morpholine group improves the compound's solubility, aiding in its distribution within biological systems. The addition of the (3-fluoro-4-morpholinophenyl) group is particularly noteworthy. The fluorine substitution enhances the compound's interaction with its target, while the morpholine moiety helps the molecule to better penetrate bacterial cell membranes. These modifications are designed to improve the compound's efficacy against bacteria that are resistant to other treatments. (S)-5-(Aminomethyl)-3-(3-fluoro-4-morpholinophenyl)oxazolidin-2-one is being explored for its potential to treat a range of bacterial infections, particularly those caused by resistant strains. Its development reflects ongoing efforts to create more effective antibiotics in the face of increasing antibiotic resistance. The compound's design aims to address the limitations of current treatments by offering a new mechanism of action and improved pharmacological properties. In addition to its potential therapeutic applications, this compound could serve as a valuable tool in pharmaceutical research. The oxazolidinone framework provides a versatile platform for developing new drugs with different mechanisms of action or targeting other bacterial or viral processes. References 2018. Synthesis and antibacterial bioactivities of cationic deacetyl linezolid amphiphiles. European Journal of Medicinal Chemistry. DOI: https://pubmed.ncbi.nlm.nih.gov/29966917 2017. Compound design guidelines for evading the efflux and permeation barriers of Escherichia coli with the oxazolidinone class of antibacterials: Test case for a general approach to improving whole cell Gram-negative activity. Bioorganic & Medicinal Chemistry Letters. DOI: https://pubmed.ncbi.nlm.nih.gov/29102393 2003. Linezolid. Pharmaceutical Substances. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-12-0041 |
| Market Analysis Reports |