Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| YURUI (Shanghai) Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.riyngroup.com | |||
 | +86 (21) 3319-1321 ex 816 +86 13032131612 | |||
 | +86 (21) 6085-3441 | |||
 | yu@riyngroup.com info@riyngroup.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Xuzhou Xbetop Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xbetop.com | |||
 | +86 (516) 8329-1698 | |||
 | +86 (516) 8329-1698 | |||
 | xbetop@gmail.com xbetop@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Sinochem Hebei Corp. | China | |||
|---|---|---|---|---|
 | www.sinochemhebei.com | |||
 | +86 (311) 8502-8964 | |||
 | xsg@sinochemhebei4.com | |||
| Chemical manufacturer since 1974 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Taoe Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.taoe-chem.com | |||
 | +86 (21) 5153-1816 | |||
 | +86 (21) 5153-1817 | |||
 | sales@taoe-chem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Henan Ouber Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oubertec.com | |||
 | +86 (371) 6532-2607 +86 18937141980 | |||
 | +86 (371) 6532-2607 | |||
 | anna.zhang@oubertec.com | |||
 | QQ Chat | |||
 | WeChat: 18937141980 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| New Fortune Industrial Co., Limited | China | |||
|---|---|---|---|---|
 | www.newfortune.top | |||
 | +86 (025) 8645-9456 | |||
 | wanqiqian0905@163.com | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Aryl compounds >> Naphthalenes |
|---|---|
| Name | 1,8-Dibromonaphtalene |
| Molecular Structure | 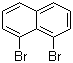 |
| Molecular Formula | C10H6Br2 |
| Molecular Weight | 285.96 |
| CAS Registry Number | 17135-74-9 |
| EC Number | 803-137-1 |
| SMILES | C1=CC2=C(C(=C1)Br)C(=CC=C2)Br |
| Solubility | 0.8791 mg/L (25 °C water) |
|---|---|
| Density | 1.8±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.688, Calc.* |
| Melting point | 105-110 °C °C (Expl.) |
| Boiling Point | 339.1±15.0 °C (760 mmHg), Calc.*, 316.45 °C |
| Flash Point | 184.7±19.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H411 Details | ||||||||||||||||||||||||||||
| Safety Statements | P264-P270-P273-P301+P317-P330-P391-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
1,8-Dibromonaphthalene is an aromatic organic compound with the molecular formula C10H6Br2. It consists of a naphthalene backbone with bromine atoms substituted at the 1- and 8-positions of the aromatic ring. This compound is a member of the halogenated naphthalene family, which has applications in a variety of fields due to the reactivity and stability imparted by the bromine atoms. The synthesis of 1,8-dibromonaphthalene typically involves bromination of naphthalene in the presence of a brominating agent, such as bromine in acetic acid. The reaction selectively introduces bromine atoms at the 1- and 8-positions of the naphthalene ring, a process that is well-established in the literature. The reaction conditions and choice of solvents are crucial in controlling the regioselectivity of the bromination, ensuring that the bromine atoms are placed at the desired positions. 1,8-Dibromonaphthalene has been studied for its use in organic synthesis. It serves as a versatile intermediate in the preparation of various organic compounds, particularly those involving the introduction of halogen atoms into aromatic systems. The presence of bromine atoms enhances the reactivity of the compound, making it a valuable precursor in the synthesis of more complex molecules, including those used in pharmaceuticals, agrochemicals, and materials science. One notable application of 1,8-dibromonaphthalene is in the development of organic electronic materials. The compound has been used in the preparation of conjugated polymers and materials for organic light-emitting diodes (OLEDs). The bromine atoms in 1,8-dibromonaphthalene can facilitate cross-coupling reactions such as the Suzuki or Stille coupling, leading to the formation of functionalized polymers with useful electronic properties. This makes it a key building block in the design of materials for use in displays, lighting, and flexible electronics. Moreover, 1,8-dibromonaphthalene has been utilized in the synthesis of various ligands and catalysts. Its halogenated structure allows for coordination with metals in catalytic processes, making it a useful compound in the development of new catalytic systems, particularly those involved in the formation of carbon-carbon bonds. The reactivity of the bromine atoms can also be exploited in the synthesis of bioactive compounds and other fine chemicals. In conclusion, 1,8-dibromonaphthalene is a halogenated aromatic compound with significant value in organic synthesis and materials science. It serves as an important intermediate in the preparation of functionalized molecules for electronic and catalytic applications. Its well-established synthesis and versatile reactivity make it a useful building block in the development of a variety of chemical and industrial products. References 1984. Crystal structure of 1,5-dibromonaphthalene and of disordered 1,8-dibromonaphthalene. Journal of Crystallographic and Spectroscopic Research, 14(6). DOI: 10.1007/bf01182146 2006. Synthesis of a Sterically Crowded Atropisomeric 1,8-Diacridylnaphthalene for Dual-Mode Enantioselective Fluorosensing. The Journal of Organic Chemistry, 71(5). DOI: 10.1021/jo0600353 2023. Extension of Highly Efficient Alcohol-promoted Rearrangement into One-pot Domino Palladium-catalyzed Carbonylation for Sterically Crowded Aromatic Six-membered Imides. Chemical Research in Chinese Universities, 39(6). DOI: 10.1007/s40242-023-3209-8 |
| Market Analysis Reports |