Online Database of Chemicals from Around the World
| BrightGene Bio-medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bright-gene.com | |||
 | +86 (512) 6255-1801 6255-1767 +86 13812696362 | |||
 | +86 (512) 6255-1799 | |||
 | kevinwan@bright-gene.com kevinwan0203@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Creative Peptides | USA | |||
|---|---|---|---|---|
 | www.creative-peptides.com | |||
 | +1 (631) 624-4882 | |||
 | +1 (631) 614-7828 | |||
 | info@creative-peptides.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shenzhen JYMed Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jymedtech.com | |||
 | +86 (755) 2661-2112 | |||
 | +86 (755) 8635-0488 | |||
 | jymed@jymedtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Zehan Biopharma Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zehanbiopharma.com | |||
 | +86 (21) 6135-0663 +86 13052117465 | |||
 | +86 (21) 6135-0662 | |||
 | sales@zehanbiopharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Nanjing Sunsure Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunsurechem.com | |||
 | +86 (25) 5853-5435 | |||
 | +86 (25) 5853-5435 | |||
 | sales@sunsurechem.com | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Tecoland Corporation | USA | |||
|---|---|---|---|---|
 | www.tecoland.com | |||
 | +1 (732) 603-9577 | |||
 | +1 (732) 906-1522 | |||
 | info@tecoland.com | |||
| Chemical distributor since 2001 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Zhejiang Boxiao Biopharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bx-biopharm.com | |||
 | +86 (571) 6328-0982 | |||
 | helena_huang@bx-biopharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | API >> Antibiotics >> Peptide drug |
|---|---|
| Name | Dalbavancin |
| Synonyms | MDL 63397 |
| Molecular Structure | 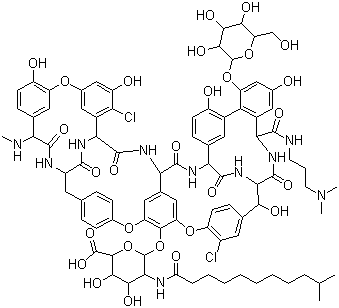 |
| Molecular Formula | C88H100Cl2N10O28 |
| Molecular Weight | 1816.69 |
| CAS Registry Number | 171500-79-1 |
| EC Number | 857-318-5 |
| SMILES | CC(C)CCCCCCCCC(=O)N[C@@H]1[C@H]([C@@H]([C@H](O[C@H]1OC2=C3C=C4C=C2OC5=C(C=C(C=C5)[C@H]([C@H]6C(=O)N[C@@H](C7=C(C(=CC(=C7)O)O[C@@H]8[C@H]([C@H]([C@@H]([C@H](O8)CO)O)O)O)C9=C(C=CC(=C9)[C@H](C(=O)N6)NC(=O)[C@@H]4NC(=O)[C@@H]1C2=C(C(=CC(=C2)OC2=C(C=CC(=C2)[C@H](C(=O)N[C@H](CC2=CC=C(O3)C=C2)C(=O)N1)NC)O)O)Cl)O)C(=O)NCCCN(C)C)O)Cl)C(=O)O)O)O |
| Solubility | Insoluble (2.0E-5 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.59±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.729, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS08;GHS09 Warning Details GHS08;GHS09 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H361-H373-H400-H410 Details | ||||||||||||||||||||
| Safety Statements | P203-P260-P273-P280-P318-P319-P391-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Dalbavancin is a second-generation lipoglycopeptide antibiotic developed to combat Gram-positive bacterial infections, particularly those caused by multidrug-resistant pathogens. Its discovery is linked to modifications of earlier glycopeptides such as vancomycin, aiming to enhance antimicrobial activity and pharmacokinetic properties. Dalbavancin is a semisynthetic derivative of the natural product A40926, which was originally isolated from Nonomuraea species. Researchers focused on structural modifications to improve stability, enhance potency against resistant bacteria, and extend the drug’s half-life, ultimately leading to its clinical approval. The United States Food and Drug Administration approved dalbavancin in 2014 for the treatment of acute bacterial skin and skin structure infections, providing a valuable alternative to conventional glycopeptides. Dalbavancin functions by inhibiting bacterial cell wall synthesis, a mechanism similar to other glycopeptides. It binds to the terminal D-Ala-D-Ala residues of peptidoglycan precursors, blocking transpeptidation and transglycosylation processes essential for cell wall formation. The result is bacterial cell lysis and death. Dalbavancin exhibits a prolonged elimination half-life of approximately 14 days, allowing for an extended dosing interval. Unlike traditional glycopeptides requiring daily administration, dalbavancin is typically administered as a single intravenous infusion or two doses one week apart, improving patient compliance and reducing hospital stays. This feature makes it particularly useful for outpatient therapy and long-term infection management. The synthesis of dalbavancin involves a semisynthetic approach based on the core A40926 structure, which is modified through selective acylation reactions. Key steps include protection and deprotection of functional groups, as well as site-specific chemical modifications to enhance its pharmacokinetic profile. The ability to optimize chemical modifications has led to improvements in drug solubility, stability, and overall therapeutic efficacy. Advances in process chemistry have made large-scale production feasible, ensuring a stable supply for clinical applications. Dalbavancin is primarily used for the treatment of acute bacterial skin and skin structure infections, particularly those caused by methicillin-resistant Staphylococcus aureus and other drug-resistant Gram-positive bacteria. Additionally, clinical trials have explored its efficacy in osteomyelitis, catheter-related bloodstream infections, and prosthetic joint infections, conditions that often require prolonged antibiotic therapy. The long-acting profile of dalbavancin makes it an attractive option for treating persistent bacterial infections, reducing the burden of frequent dosing and hospitalization. Research is ongoing to assess its potential in combination therapies and expand its indications beyond skin infections. The widespread use of dalbavancin is limited by its high cost, which can restrict accessibility despite its clinical advantages. However, its effectiveness against multidrug-resistant bacteria and its ability to streamline antibiotic therapy continue to drive interest in its application. With increasing antibiotic resistance posing a global challenge, dalbavancin remains a crucial component of modern antimicrobial strategies. Future studies will likely explore its role in combination regimens and broader therapeutic applications, reinforcing its importance in infectious disease management. References 1999. In-vitro and in-vivo antibacterial activity of BI 397, a new semi-synthetic glycopeptide antibiotic. The Journal of antimicrobial chemotherapy, 44(2). DOI: 10.1093/jac/44.2.179 2024. Real-life experience with IV dalbavancin in Canada; results from the CLEAR (Canadian LEadership on Antimicrobial Real-life usage) registry. Journal of Global Antimicrobial Resistance, 38. DOI: 10.1016/j.jgar.2024.06.002 2024. A comprehensive study on the identification and characterization of degradation products of lipoglycopeptide Dalbavancin using LC and LC‐HRMS/MS. Journal of peptide science : an official publication of the European Peptide Society, 30(10). DOI: 10.1002/psc.3608 |
| Market Analysis Reports |