Online Database of Chemicals from Around the World
| Highpharm Medchem (Ningbo) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.highpharm.com | |||
 | +86 (574) 5566-3958 | |||
 | +86 (574) 5566-3960 | |||
 | highpharm@highpharm.com lisayang925@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Shanghai Zehan Biopharma Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zehanbiopharma.com | |||
 | +86 (21) 6135-0663 +86 13052117465 | |||
 | +86 (21) 6135-0662 | |||
 | sales@zehanbiopharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhuhai Hairuide Bioscience and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhhairuide.com | |||
 | +86 (756) 778-9199 +86 13326687259 +86 13326687289 +86 15913232160 | |||
 | +86 (756) 778-9099 | |||
 | sales@zhhairuide.com zhengyanhong@zhhairuide.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Beri Pharma Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhberi.com | |||
 | +86 19500796935 | |||
 | elena@zhberi.com | |||
 | Skype Chat | |||
 | WeChat: 19500796925 | |||
 | WhatsApp:+8619500796925 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Digestive system medication >> Inhibition of gastric acid secretion |
|---|---|
| Name | Ilaprazole |
| Synonyms | 2-[[(4-Methoxy-3-methyl-2-pyridinyl)methyl]sulfinyl]-6-(1H-pyrrol-1-yl)-1H-benzimidazole; IY 81149 |
| Molecular Structure | 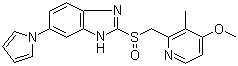 |
| Molecular Formula | C19H18N4O2S |
| Molecular Weight | 366.44 |
| CAS Registry Number | 172152-36-2 |
| SMILES | CC1=C(C=CN=C1CS(=O)C2=NC3=C(N2)C=C(C=C3)N4C=CC=C4)OC |
| Solubility | 5.923 mg/L (25 °C water) |
|---|---|
| Density | 1.4±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.710, Calc.* |
| Melting point | 271.46 °C |
| Boiling Point | 651.0±65.0 °C (760 mmHg), Calc.*, 626.01 °C |
| Flash Point | 347.5±34.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
| SDS | Available |
|
The discovery of Ilaprazole represents a significant breakthrough in the field of gastroenterology and acid-related disorders. Ilaprazole, a proton pump inhibitor (PPI), was developed through extensive research aimed at improving upon existing treatments for conditions such as gastroesophageal reflux disease (GERD), peptic ulcers, and gastritis. Scientists synthesized Ilaprazole by modifying the chemical structure of earlier PPIs, enhancing its efficacy and pharmacokinetic properties. Ilaprazole is widely prescribed for the management of GERD, a condition characterized by the reflux of stomach acid into the esophagus, causing heartburn and esophageal damage. By inhibiting gastric acid secretion, Ilaprazole alleviates symptoms and promotes healing of esophageal tissue, providing relief to patients suffering from GERD.Ilaprazole is effective in the treatment and prevention of peptic ulcers, including gastric ulcers and duodenal ulcers. Ilaprazole is utilized in the management of gastritis, an inflammation of the stomach lining often associated with excessive gastric acid secretion. Ilaprazole is included in combination therapy regimens for the eradication of Helicobacter pylori, a bacterium implicated in the pathogenesis of peptic ulcers and gastric cancer. Combined with antibiotics, Ilaprazole helps suppress acid production and creates an environment unfavorable for bacterial growth, facilitating the eradication of H. pylori infection. In hospitalized patients at risk of stress ulcers, Ilaprazole is used for prophylaxis to prevent ulcer formation. Ilaprazole may be prescribed for the management of non-ulcer dyspepsia, a common digestive disorder characterized by symptoms such as upper abdominal pain, bloating, and discomfort. Its acid-suppressing effects alleviate dyspeptic symptoms and improve overall gastrointestinal function in affected individuals. References 1999. Identification of IY81149 and its metabolites in the rat plasma using the on-line HPLC/ESI mass spectrometry. Archives of Pharmacal Research, 22(2). DOI: 10.1007/bf02976545 2023. Effectiveness of combined proton pump inhibitors and posaconazole prophylaxis against invasive fungal infections in patients with hematologic malignancies: a retrospective study. International Journal of Clinical Pharmacy, 46(1). DOI: 10.1007/s11096-024-01841-8 2000. A comparative pharmacodynamic study of IY-81149 versus omeprazole in patients with gastroesophageal reflux disease. Clinical pharmacology and therapeutics, 68(3). DOI: 10.1067/mcp.2000.109155. |
| Market Analysis Reports |