Online Database of Chemicals from Around the World
| Shanghai Chengjie Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyfhx.com | |||
 | +86 (21) 6489-6788 | |||
 | +86 (21) 3392-1761 | |||
 | shyfhx@aliyun.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Monils Chemical Engineering Science & Technology (shanghai) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ilschem.cn | |||
 | +86 (21) 3822-8895 | |||
 | +86 (21) 3822-8591 | |||
 | heqing@ilschem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Zhejiang Arts & Crafts Imp & Exp.co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjartspharm.com | |||
 | +86 571 87238903 | |||
 | jocelynpan@zjarts.com | |||
| Chemical distributor since 1992 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| Shanghai Cainorise Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cainorise.com.cn | |||
 | +86 (21) 3408-0216 2950-8524 +86 15921434758 | |||
 | sales@cainorise.com.cn | |||
| Chemical manufacturer | ||||
| abcr GmbH | Germany | |||
|---|---|---|---|---|
 | www.abcr.de | |||
 | +49 (721) 950-610 | |||
 | +49 (721) 950-6180 | |||
 | info@abcr.de | |||
| Chemical manufacturer | ||||
| IOLITEC Ionic Liquids Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.iolitec-usa.com | |||
 | +1 (205) 348-2831 | |||
 | +1 (205) 348-3510 | |||
 | info@iolitec-usa.com | |||
| Chemical manufacturer since 2002 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ionic liquid |
|---|---|
| Name | 1-Butyl-3-methylimidazolium trifluoromethansulfonate |
| Molecular Structure | 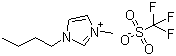 |
| Molecular Formula | C8H15N2.CF3O3S |
| Molecular Weight | 288.29 |
| CAS Registry Number | 174899-66-2 |
| EC Number | 678-094-3 |
| SMILES | CCCCN1C=C[N+](=C1)C.C(F)(F)(F)S(=O)(=O)[O-] |
| Density | 1.292 |
|---|---|
| Melting point | 16 °C |
| Refractive index | 1.434 |
| Hazard Symbols |    GHS06;GHS07;GHS09 Danger Details GHS06;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H315-H319-H335-H411 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P273-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
1-Butyl-3-methylimidazolium trifluoromethanesulfonate (BMIM OTf) is an ionic liquid with the molecular formula C9H15F3N2O3S. The substance belongs to a class of organic salts that remain liquid at relatively low temperatures, and has unique properties that have a wide range of applications in science and industry. The discovery and development of BMIM OTf was part of a broad study of ionic liquids that began in the mid-20th century. Scientists were exploring alternatives to traditional organic solvents that could offer advantages such as low volatility, non-flammability, and high thermal stability. BMIM OTf was synthesized to exploit these properties for a variety of applications. Its synthesis involves the reaction of 1-butyl-3-methylimidazolium chloride (BMIM Cl) with silver trifluoromethanesulfonate (AgOTf), resulting in the exchange of chloride ions with trifluoromethanesulfonate ions to form BMIM OTf. In this reaction, BMIM Cl reacts with AgOTf to form BMIM OTf and silver chloride (AgCl) as a byproduct. The reaction usually takes place in a suitable solvent, followed by purification to isolate the desired ionic liquid. The most important application of BMIM OTf is as a green solvent in chemical processes. Traditional organic solvents are volatile and toxic, posing risks to the environment and health. BMIM OTf, on the other hand, is non-volatile and recyclable, reducing the environmental impact of chemical reactions. It is used in organic synthesis, catalysis, and extraction processes to improve reaction efficiency and selectivity. BMIM OTf is widely used in electrochemical applications due to its high ionic conductivity and stability. It can be used as an electrolyte in batteries, fuel cells, and supercapacitors, improving their performance and efficiency. Its wide electrochemical window and low viscosity make it a first choice for the development of advanced energy storage and conversion devices. In catalysis, BMIM OTf can be used as both a solvent and a catalyst. It is able to dissolve a wide range of organic and inorganic compounds, making it a versatile medium for catalytic reactions. BMIM OTf is used in homogeneous and heterogeneous catalysis to promote reactions that are difficult to achieve with traditional solvents. It helps to increase reaction rates, yields, and selectivity, and is of great value in sustainable chemical synthesis. BMIM OTf is used in extraction and separation processes such as liquid-liquid extraction and gas absorption. Its unique solvation properties allow for the selective extraction of specific compounds from a mixture, thereby increasing the efficiency and selectivity of the separation process. It is used for the purification of drugs, the extraction of metals, and the separation of organic compounds. In the pharmaceutical industry, BMIM OTf is used for drug formulation and delivery. Its unique properties enable poorly soluble drugs to be solubilized and stabilized, thereby increasing their bioavailability. In addition, its use in drug synthesis can lead to more efficient and selective reactions, aiding in the development of new drugs. BMIM OTf is also used in materials science to synthesize and modify materials with specific properties. It plays an important role in the preparation of nanoparticles, polymers, and other advanced materials. BMIM OTf can act as a template or stabilizer during the synthesis of nanomaterials, affecting their size, shape, and stability. References 2023. Tutorial for thermal analysis of ionic liquids. Journal of Thermal Analysis and Calorimetry. DOI: 10.1007/s10973-023-12439-z 2022. Transformations of Imidazolium Ionic Liquids under the Influence of 60Co Gamma Radiation. Doklady Physical Chemistry. DOI: 10.1134/s0012501622040017 2008. A one-pot synthesis of functionalized ethyl 1,3-thiazole-5-carboxylates from thioamides or thioureas and 2-chloro-1,3-dicarbonyl compounds in an ionic liquid. Monatshefte für Chemie - Chemical Monthly. DOI: 10.1007/s00706-008-0065-7 |
| Market Analysis Reports |